The Na, K-pump or Na/K-ATPase actively transports Na and K ions across mammalian cell membranes to establish and maintain the characteristic trans membrane gradients of Na and K ions. This function underlies essentially all of mammaliancell physiology.Forexample, in the kidney, the Na, K-pump controls body Na and K balance, extracellular volume and blood pressure. In the heart the Na, K-pump controls myocyte Ca balance and cardiac contractility. The Na, K-pump is the receptor of digitalis steroids used to treat heart failure. Na/K-ATPase is a membrane protein and consists of a catalytic α subunit with ten trans-membrane segments, and a single trans-membrane glycosylated β subunit, required for stabilization. Na,K-ATPase is regulated by FXYD proteins which are auxiliary subunits. There are four isoforms of α (1-4) and three isoforms of β expressed in a tissue-specific fashion α1 is the “housekeeping” isoform. α2 is expressed in heart and other muscle and plays a key role in maintenance of blood pressure and cardiac function.
The Na, K-ATPase is a member of the P-type ATPase family of cation pumps that use the free energy of hydrolysis of ATP to actively transport cations against their electrochemical gradients. Other P-type ATPases include sarcoplasmic reticulum Ca-ATPase, gastric cell membrane H/K-ATPase, plasma membrane Ca-ATPase, plant cell membrane H-ATPase, heavy metal-dependent ATPases etc., with selectivity for the other cations.

P-type ATPases have a common kinetic mechanism, which involves covalent phosphorylation of an active site aspartate residue by ATP, an E1P E2P conformational change coupled to cation movement, hydrolysis of the phosphoenzme and an E2-E1 conformational change to complete the cycle. Crystal structures of sarcoplasmic reticulum Ca-ATPase, and native renal Na,K-ATPase, published recently, illuminate the basic mechanism of active cation transport. Neverthless insights into crucial features of Na,K pump structure, function, regulation and pharmacology are lacking.
Structure- crystalization and modeling
We have expressed Na,K-ATPase in the methanotrophic yeast, Pichia pastoris, and purified the protein to homogeneity in a single step (Strugatsky et. al., 2003; Cohen et. al, 2005; Haviv et. al., 2007). About 1-2mg of pure, stable and functional α β complexes in a non-ionic detergent can be prepared conveniently. Initially porcine and human α1/β1 were purified . An essential feature is that specific interactions with phosphatidyl serine (PS) are required to stabilize the protein, probably at a site near the α β subunit interface. More recently the human 2 β1 isoform complex has been expressed, purified and stabilized (Lifshitz et. al, 2007). α2 is unstable compared to α1 due to weaker phospholipid-protein interactions and must be stabilized by a combination of PS/cholesterol. Other isoforms α β subunits are now being expressed.
Crystalization trials are being carried out. If suitable crystals are obtained it is hoped that these will lead to determination of structure of different conformations of the protein, mutants, isoforms, and complexes with FXYD proteins (see below).
The β subunit plays an essential role as a chaperone of α, and is known also to play an important role in cell-cell adhesion. However, its structure has not been well defined. We have used Fold Recognition methods to predict that the extracellular domain has an Imunoglobulin-like fold and consists of two lobes (Fig. 2). This concept has interesting implications for the physiological role of the β subunit. We are now attempting to express and purify these putative lobes of the protein (Dinitz and Karlish, unpublished).
Function – E1-E2 conformational changes
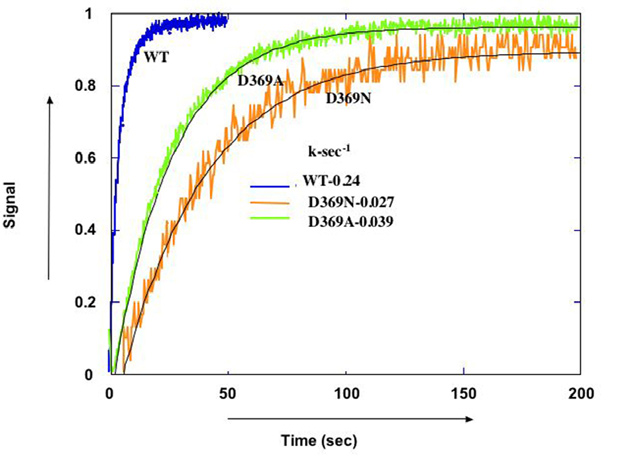
Crystal structures have shown that the essence of E1-E2 conformational changes is a movement of cytoplasmic domains (N, P, and A) coupled to movement of trans-membrane segments, which mediates the cation transport. However, it is not known what triggers the conformational changes. Previously we hypothesized that changes in charge on active site aspartate (D369) upon phosphorylation, are the trigger (Strugatsky et.al, 2003). We have now utilized purified fluorescein-labeled recombinant Na,K-ATPase (see Karlish, 1980) to look at effects of charge of D369 on conformational changes, by comparing wild-type and charge neutralized mutants (D369N and D369A). Steady-state and transient kinetics of fluorescence changes show that the charge on D369 is indeed a crucial feature (Belogus and Karlish, unpublished) ( Fig. 2).
In another approach we have utilized the technique of Fe-catalyzed oxidative cleavage , developed to analyze spatial organization of proteins around specifically bound Fe (reviewed Karlish, 2003), to investigate divalent metal sites in the Na,K-ATPase expresed in Pichia Pastoris (Strugatsky et. al.,2005). This suggested that two Mg ions are bound to the protein in the complex with ATP-Mg, one in the P-domain (D710) and a second in the N domain (D443).
Regulation- FXYD proteins (with Prof. Haim Garty, Dept. Biological Chemistry)
FXYD proteins are a group of seven short single span transmembrane proteins termed after the invariant motif FXYD in their extracellular domain. FXYD proteins act as tissue-specific regulatory subunits, which adjust the kinetics properties of the Na+, K+-pump to the needs of the particular cell type or physiological state (reviewed in Garty and Karlish, 2006). We have investigated intensively the functional effects and structural interactions of FXYD 1, 2, 4 and 5 expressed in in mammalian cells and Xenopus oocytes. Most recently we have been focussing on interactions of purified FXYD1 (phospholemman, PLM). FXYD1 regulates the Na+, K+-pump in cardiac and skeletal muscle. PLM has PKA and PKC phosphorylation sites and responds to -adrenergic and other hormonal signals.
To go to Interaction with the Na click here 
FYYD1 has been expressed in Pichia Pastoris and reconstituted with purified human α1/β1 and α2/β1 complexes to produce α1/β1/FXYD1 and α2/ β1/FXYD1 complexes (Lifshitz et. al., 2006;Lifshitz et. al., 2007). The functional properties of α1/β1/FXYD1 (phosphorylated or not at Ser68) have been characterized. A striking feature of the α1/β1/FXYD1 and α2/β1/FXYD1 complexes is that they are highly thermally stabilized by comparison with α1/β1 and α2/β1 complexes (Lifshitz et. al., 2007). FXYD1 stabilizes the phosphatidyl serine-α/β interaction. More recently we have expressed FXYD1 in E.Coli purified the protein and reconstituted α1/β1/FXYD1 complexes (Lifshitz and Karlish, unpublished, in collaboration with the Weizmann Institute Proteomics Center). The purified α1/β1/FXYD1 complex will be used for crystalization trials and detailed functional characterization. Similar experiments with FXYD2 have been initiated.
Pharmacology- an α2-selective cardiac glycoside (CG)?
Plant-derived digitalis steroids have been used for over two hundred years to increase the force of contraction of the heart (positive inotropy), but they are dangerous drugs and can induce fatal arrhythmias. In addition it is known that digitalis-like steroids are produced in mammals in a manner similar to steroid hormones, and are intimately involved in regulation of blood pressure and cardiac hypertrophy. Thus, there is great interest in the mechanism of action of endogenous CG’s and, development of safer CG’s. One way to reduce digitalis toxicity could be to develop an inhibitor selective for the α2 isoform. α1,α2 α3 isoforms are all expressed in humans hearts, but α1 is the predominant isoform. We are utilizing the human α1β1 and α2β1 complexes purified from P.pastoris membranes to try and develop an α2-selective inhibitor. This involves a combination of biochemical screening, synthetic chemistry and molecular modeling.
Selected publications
Karlish, S.J. (1980) Characterization of conformational changes in (Na,K) ATPase labeled with fluorescein at the active site. J Bioenerg Biomembr, 12, 111-136. Strugatsky, D., Gottschalk, K.E., Goldshleger, R., Bibi, E. and Karlish, S.J. (2003) Expression of Na+,K+ATPase in Pichia pastoris: analysis of wild type and D369N mutant proteins by Fe2+-catalyzed oxidative cleavage and molecular modeling. J Biol Chem, 278, 46064-46073. Karlish, S.J. (2003) Investigating the energy transduction mechanism of P-type ATPases with Fe2+-catalyzed oxidative cleavage. Ann N Y Acad Sci, 986, 39-49. Cohen, E., Goldshleger, R., Shainskaya, A., Tal, D.M., Ebel, C., le Maire, M. and Karlish, S.J. (2005) Purification of Na+,K+-ATPase expressed in Pichia pastoris reveals an essential role of phospholipid-protein interactions. J Biol Chem, 280, 16610-16618. Strugatsky, D., Gottschalk, K.E., Goldshleger, R. and Karlish, S.J. (2005) D443 of the N domain of Na+,K+-ATPase interacts with the ATP-Mg2+ complex, possibly via a second Mg2+ ion. Biochemistry, 44, 15961-15969. Garty, H. and Karlish, S.J. (2006) Role of FXYD proteins in ion transport. Annu Rev Physiol, 68, 431-459. Lifshitz, Y., Lindzen, M., Garty, H. and Karlish, S.J. (2006) Functional interactions of phospholemman (PLM) (FXYD1) with Na+,K+ATPase. Purification of alpha1/ beta1/PLM complexes expressed in Pichia pastoris. J Biol Chem, 281, 15790-15799. Haviv, H., Cohen, E., Lifshitz, Y., Tal, D.M., Goldshleger, R. and Karlish, S.J. (2007) Stabilization of Na(+),K(+)ATPase purified from Pichia pastoris membranes by specific interactions with lipids. Biochemistry, 46, 12855-12867. Lifshitz, Y., Petrovich, E., Haviv, H., Goldshleger, R., Tal, D.M., Garty, H. and Karlish, S.J. (2007) Purification of the human alpha2 Isoform of Na,K-ATPase expressed in Pichia pastoris. Stabilization by lipids and FXYD1. Biochemistry, 46, 14937-14950.