Designer cellulosomes as a platform for processing biomass-to-biofuels
Cellulose is a simple but incredibly resilient polymer, composed solely of the common sugar glucose.
Together with other related plant cell wall carbohydrates, cellulose comprises the most abundant source of renewable carbon and energy on Earth.
If we are to circumvent the current dependence of modern society on fossil fuels, namely petroleum, and its resultant political and social fallout, we must tap into the renewable cellulose resource as a supplement/replacement for our oil-based economy.
No efficient chemical process is available for this conversion, and the major bottleneck in the biotechnological conversion of cellulose to useful sugars and biofuels is the inefficient degradation of this resistant plant polymer by natural microbial enzymes.
Our work in this area aims at the application of innovative experimental approaches towards the generation of artificial enzyme systems – designer cellulosomes – for efficient degradation of cellulose to glucose, for subsequent cost-effective conversion to ethanol.
The AERI Project
Engineering Bioreactors
AERI
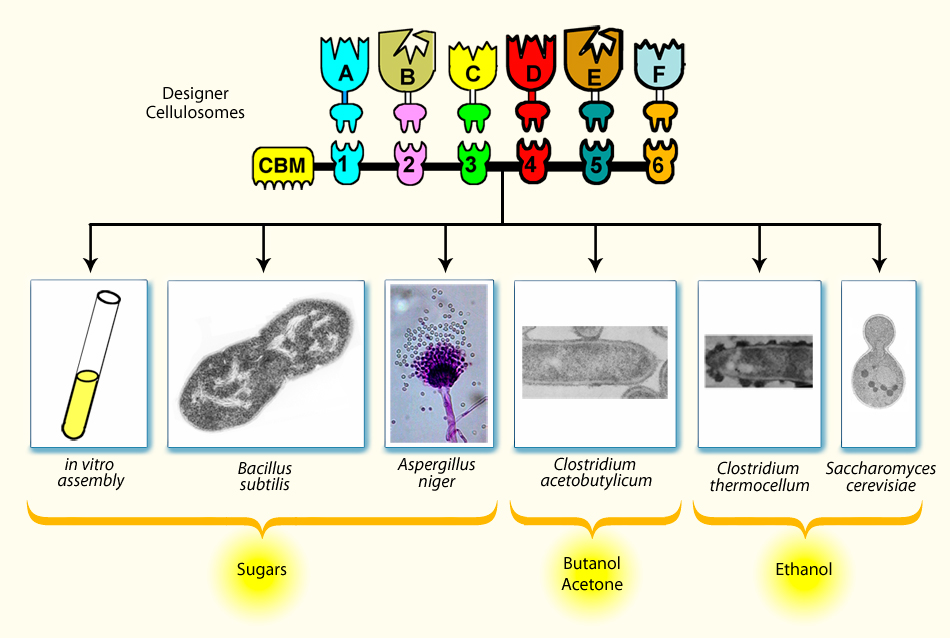
Engineering of Cellulolytic Bioreactors
Engineering potent cellulolytic microbes for the production of desired end products. Genes encoding for cellulases and/or designer cellulosome components can be cloned into a desired bacterial or fungal host cell, and the secreted proteins can be overexpressed for the degradation of cellulosic biomass in an industrial reactor (in vitro assembly). Alternatively, the genes can be cloned into a suitable bacterial, fungal or yeast host, and the transformed cell, with either de novo or improved cellulose-degrading capacity, can be grown directly on cellulosic biomass to produce a desired end product.
We plan to exploit the designer cellulosome approach in order to establish effective medium- and high-throughput procedures for obtaining improved cellulases and cellulosomes that exhibit enhanced degradative properties on cellulosic substrates. The proposal brings together three Weizmann Institute scientists from the same department:

Ed Bayer, co-discoverer of the cellulosome and designer cellulosome concepts, who has worked in the area of cellulose degradation for the past 25 years

Gideon Schreiber, a leading figure in the field of biophysical studies of protein-protein interactions, and protein-interface design

Dan Tawfik, a leading expert in enzyme evolution and the development and application of directed evolution technologies.
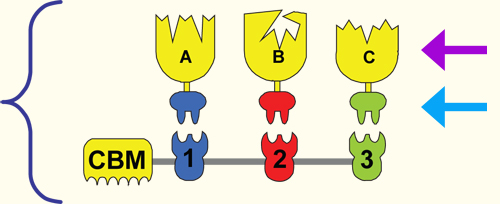
1. Development of designer cellulosomes
2. Using MTP and HTP to develop specific cohesin-dockerin interactions and alternatives
3. Directed Evolution: Cellulase and cellulosome improvement
The improved cellulases and designer cellulosomes derived from this study will help us understand and overcome obstacles that hamper efficient enzymatic degradation of cellulose and will provide novel efficient enzymes and enzyme complexes that will facilitate biomass conversion.
Directed Evolution Of Cellulolytic Enzymes
Cellulose biodegradation by cellulases and cellulosomes is important in several agricultural and waste treatment processes, and could be widely used to produce sustainable biobased products and bioenergy to replace depleting fossil fuels. One of the most important and difficult technological challenges is to overcome the recalcitrance of natural lignocellulosic materials, which must be enzymatically hydrolyzed to produce fermentable sugars. Cellulases are relatively costly enzymes, and a significant reduction in cost will be important for their commercial use in biorefineries. Thermostable enzymes are gaining wide industrial and biotechnical interest due to the fact that they are more stable and thus generally better suited for harsh process conditions. Thermostable enzymes in the hydrolysis of lignocellulosic materials have several potential advantages: higher specific activity (decreasing the amount of enzyme neewould be to apply in using the directed evolution approach, in which the improved enzymes or ones with new properties are selected or screened after random mutagenesis and/or molecular recombination. We are currently constructing random libraries of C. thermocellum cellulosomal enzymes, in order to select for improved enzymatic and stability properties. The enzymes will be re-incorporated into designer cellulosomes in order to produce superior synergistically acting enzyme complexes.



