Abstract
Dunaliella is a unicellular green alga that can tolerate high salinity and iron deprivation, two major stresses for photosynthetic organisms. Our laboratory’s aims are: (i) to identify proteins associated with stress resistance and to clone their corresponding genes, (ii) to clarify the structural basis enabling proteins/enzymes to function at high salt concentrations, (iii) to utilize genes from Dunaliella to generate transgenic plants with enhanced salinity tolerance.
A differential proteomic analysis revealed that salt stress upregulates in Dunaliella key enzymes in photosynthetic carbon assimilation and energy production. We developed procedures for tagging and resolution of plasma membrane proteins that enabled to identify salt-regulated proteins in Dunaliella plasma membranes. Surface-exposed plasma membrane proteins that are acidic and consist of two or three repeating units, properties believed to be associated with salt resistance.
Dunaliella salina adapts to iron deficiency by up-regulation Fe uptake and by remodeling of their photosynthetic system. Four major proteins, including two transferrins and a multicopper ferroxidase are induced at limiting iron in the plasma membrane. These proteins assemble to create an iron-binding complex at the plasma membrane surface, which binds, occludes and internalizes large amounts of Fe3+ ions. A novel chlorophyll a/b binding protein is induced in photosysyem I (PS-I) leading to enlargement of PS-I antenna and rebalancing of the energy distribution between PS-I and PS-II.
The findings reveal how organisms can adapt to extreme salinity or iron deprivation and provide potential molecular elements for enhancing salinity tolerance in crop plants.
Introduction
Dunaliella is a unicellular green alga that is unique in its ability to adapt to some of the harshest environments known on earth. Most Dunaliella species tolerate extreme salinities and will survive even in The Dead Sea, D. acidophila has adapted to the most acidic habitats on earth and grows optimally at pH 0-1, D. bardawil tolerates exceptionally high irradiance and D. antarctica thrives at subzero temperatures and is resistant to freezing. The special interest in Dunaliella is more than a scientific curiosity: D. bardawil in the richest known source of natural beta-carotene and is cultured in NBT Eilat for commercial production of this important chemical. Dunaliella is also a native pool for stress-related genes that may be utilyzed for generation of robust crop plants to enhance agricultural productivity.
Specific topics
Salt-resistance proteins
In a recent analysis of soluble salt-induced proteins, we have shown that Dunaliella responds to salt stress by upregulation of major metabolic pathways associated with photosynthetic carbon assimilation and energy utilization (Liska et al., 2004).
Our main effort is to identify the changes in the plasma membrane proteome under salt stress. We developed procedures for tagging plasma membrane proteins, their resolution on Blue-Native gels, followed by mass spectrometry analysis and sequence homology searches. Salt-induced proteins in the plasma membrane include a unique
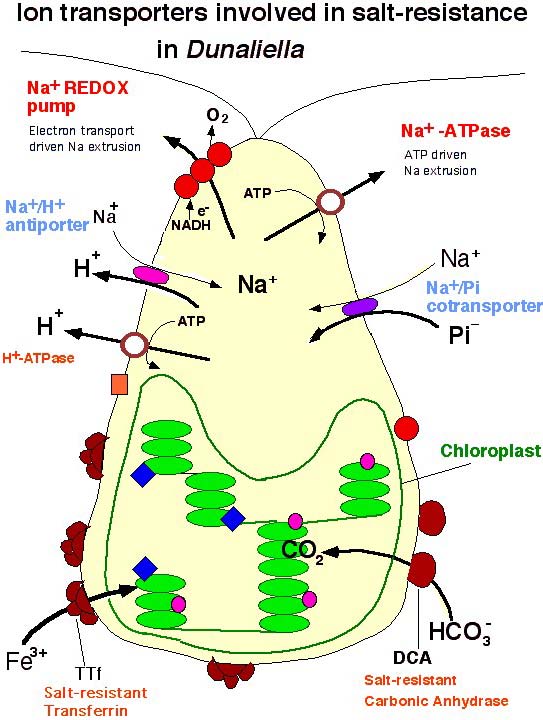
Life Science Open Day | 2006 | Weizmann Institute of Science transferrin mediating uptake of Fe3+ ions, two salt-resistant carbonic anhydrases (studied by A. Zamir), a redox-driven Na+ pump and a nitrate and ammonium transporters. The results established that iron and CO2 acquisition are major rate limiting steps for survival in hypersaline environments.
Genes of selected salt-induced proteins, such as a salt and acid-resistant H+-ATPase, have been introduced into crop plats for generation of salt-resistant crops as part of a MAGNET program with plant seed and tissue-culture companies (M. Weiss).
Na+ transport
Iron acquisition
D. salina over-expresses 4 major plasma membrane proteins under Fe deprivation: two different transferrins, a multi-copper ferroxidase and an unknown 130 kDa protein. These proteins form an iron-binding complex at the cell surface which enables binding and occlusion of 106-107 Fe3+ ions per cell, acting as a high affinity trap for iron. The bound iron is subsequently internalized into acidic vacuoles (J. Paz). This confers Dunaliella a high competitive potential for survival at limiting Fe. (ii) Remodeling of PS-I: We found that the photosynthetic system of Dunaliella is being remodeled under iron limitation: we identified and cloned a novel chlorophyll a/b binding protein, that is induced in photosystem I (PS-I) under Fe deprivation, leading to enlargement of the light harvesting antenna of PS-I (Fig 2). These studies demonstrated a new strategy to cope with iron deprivation that may be common to other photosynthetic organisms (T. Varsano, S. G. Wolf).
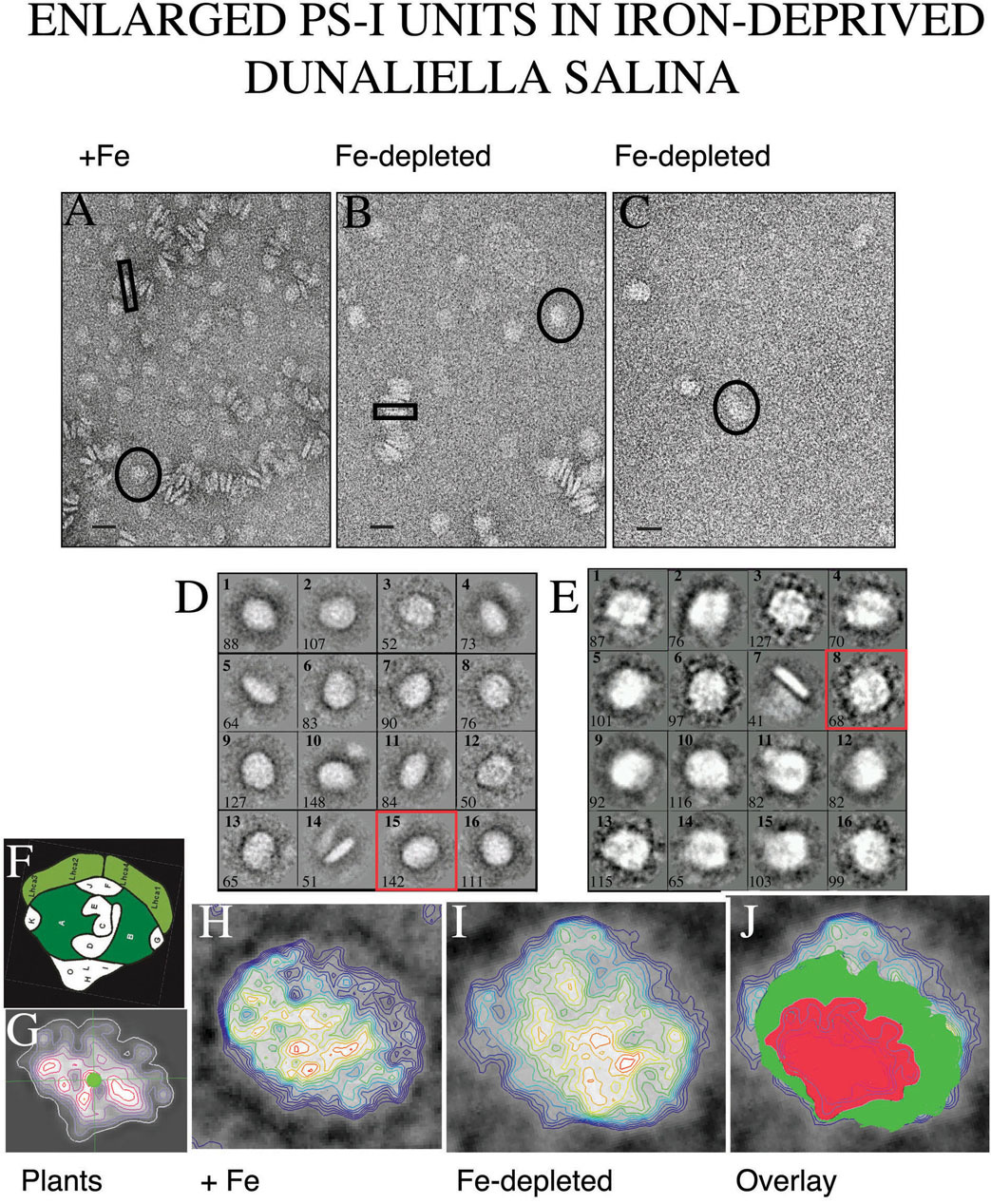
U., 2006).
Salt-resistant proteins are acidic and contain internal repeats.
Selected Publications
Pick, U. (1998), Dunaliella- a model extremophilic alga, Israel J. Plant Sci. 46, 131-140.
Katz, A. and Pick U. (2001) Plasma membrane electron transport coupled to Na+ extrusion in the halotolerant alga Dunaliella. Biochim. Biophys. Acta, 1504, 423-31.
Pick, U. (2001) Adaptation of the halotolerant alga Dunaliella to high salinity. In: Salinity, environment, plants, molecules. Lauchli, A. and Luttge, U. eds., Kluwer Acad. pp. 97-112.
Weiss M, Haimovich G & Pick U (2001) Phosphate and sulfate uptake in the halotolerant alga Dunaliella are driven by Na+symport mechanism. J. Plant Physiol. 158: 1519-1525
Pick U. (2002) Adaptation of the halotolerant alga Dunaliella to high salinity. In: Salinity: environment-plants-molecules (Läuchli A. & Lüettge U eds) Kluwer Acad. Pub./ Dordrecht the Netherlands pp. 97-112.
Zchut S., Keren N., Ohad I. & Pick, U. (2002) Cold-acclimation protects photosystem II in the halotolerant alga Dunaliella against freezing , J. Plant Physiol, 160(2), 185-192.
Varsano, T, Kaftan, D. Pick, U. (2003) Effect of iron deficiency on thylakoid membrane structure and composition in the alga Dunaliella salina J. Plant Nut. 26 (10/11) 2043-56.
Schwarz, M., Zamir, A. Pick, U. (2003) Biochemical characterization of TTf- the triplicated transferrin-like protein from the halotolerant alga Dunaliella salina. J. Plant Nut., 26(10/11) 2081-2092.
Zcut S., Weiss M and Pick, U. (2003) A glycine-rich RNA-binding protein is induced at low temperature in the halotolerant alga Dunaliella salina. J. Plant. Physiol., 160/11. 1375-1384).
Schwarz, M., Sal-Man, N., Zamir, A. Pick, U. (2003) A transferrin-like protein that does not bind iron is induced by iron deficiency in the alga Dunaliella salina. Biochim.Biophys. Acta, 1649, 190).
200.
Pick, U. (2003) The respiratory inhibitor antimycin A specifically binds Fe-III ions and mediates utilization of iron by the halotolerant alga Dunaliella. Biometals, 17,79-86.
Liska, AJ, Shevchenko A, Pick U, Katz A (2004) Elevated Photosynthesis and Redox Energy Production Contribute to Salinity Tolerance in Dunaliella as Revealed by Homology-Based Proteomic Analysis. Plant Physiol. 136:1-12.
Varsano T, Wolf S.G. and Pick U. (2006) A novel Chlorophyll a/b binding protein is induced in PS-I in PS-I by Iron deficiency in the alga Dunaliella salina. J. Biol Chem. (in press).
