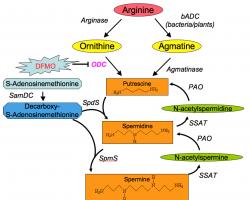
Polyamine synthesis occurs in the cytoplasm of cells from all tissues. Polyamines are synthesized from two amino acids: L-methionine and L-ornithine (an amino acid that is not found in proteins, that is produced as part of the urea cycle).
In mammalian cells, putrescine is formed by decarboxylation of ornithine, a reaction catalyzed by the enzyme ornithine decarboxylase (ODC). Ornithine is available from the plasma and can also be formed within the cell from arginine by the action of arginase.
Synthesis of spermidine and spermine require the action of two enzymes: first, the S-adenosyl-methionine decarboxylase (AdoMetDC) for the synthesis of the aminopropyl donor; and second, a transferase enzyme (spermidine synthase or spermine synthase) which catalyze the transfer of the aminopropyl group to the primary amine groups of putrescine or spermidine, respectively.
Spermine and spermidine are also regulated by the presence of a distinctive interconversion pathway, where they are acetylated (by spermidine/spermine acetyltransferase SSAT), and oxidized (by polyamine oxidase POA) back to putrescine.
The third level of regulating polyamine metabolism is the terminal degradation of polyamines: oxidation of primary (terminal) amino groups produces polyamine derivatives that cannot be cycled back into polyamines. Polyamines are oxidized by variety of oxidases with different modes of action and co-factor requirements.
In light of their fundamental significance, it is unsurprising that the intracellular level of polyamines has to be maintained within very narrow limits. Decreases of polyamine levels interfere with cell growth, leading to G1 arrest in S. cerevisiae and to embryonic lethality in mice. Abnormally high levels of polyamines appear to be toxic, causing apoptosis in mammalian cells. Polyamine content is increased in many cancers arising from epithelial tissues, such as skin and colon.