Publications
2026
-
51.(2026) BioRxiv. Abstract
Single-cell phenotypic diversity underlies fundamental bacterial behaviors. Resolving this heterogeneity at scale while maintaining sensitive detection remains a central challenge. Here, we address this gap by combining the strengths of single-molecule fluorescence in situ hybridization (smFISH) and transcriptome-wide sequencing. We present par2FISH, a framework for discovering transcriptionally defined subpopulations across conditions by multiplexing tens of thousands of smFISH reactions. Through comparative single-cell transcriptomics in Escherichia coli and Salmonella, we uncover conserved and species-specific patterns of heterogeneity, including mutually exclusive states, coordinated metabolic specialization, and differential activation of virulence programs. We further obtain complete transcriptomes for selected subpopulations using marker-based cell sorting and RNA-sequencing (FACS-RNA-seq). This integrated strategy paves the way for exposing bacterial phenotypic landscapes and the environmental, physiological, and evolutionary factors that shape them.
2025
-
50.(2025) Nature Communications. 16, 1, 6755. Abstract[All authors]
Salmonella Typhi (S. Typhi), the causative agent of typhoid disease, remains a major public health concern. Owing to the human-restricted nature of S. Typhi, studies of typhoid pathogenesis in animal models are limited to a murine non-typhoidal pathogen. More recently, human challenge models have been conducted, providing insight into immune correlates of infection outcomes, which are still incompletely understood. Here, we performed an integrated single-cell analysis of immune responses from the human S. Typhi challenge model and mouse model of typhoid disease, to associate biological mechanism with human infection outcome. Most prominent, we revealed immune subsets with a hypoxia-related signature in the blood of individuals who developed disease in the human challenge model. This signature was also evident in the mouse model in activated macrophages infiltrating into the Peyers patches, but not during infection with a mutant strain impaired for gut invasion. We further identified hypoxia-related signature as a general immune correlate of disease outcome in other infection-and inflammatory-related diseases. Collectively, we identified a hypoxia-associated immune signature that correlates with disease outcomes in humans. Using a mouse model, we demonstrated that this signature is driven by bacterial invasion to the Peyers patches, implicating a causal role in the pathogenesis of typhoid fever.
-
49.(2025) Manual of Molecular Microbiology. McAdam A. J., Wolk D. M., Dekker J. P., Carroll K. C. & Hayden R. T.(eds.). p. 137-143 Abstract
Transcriptomics, a fundamental area within molecular genetics, focuses on the abundance and profile of RNA transcripts within a given cell, and how changes in their expression govern the complex cellular activities that maintain equilibrium and enable adaptation to fluctuating environmental conditions. Technological improvements to RNA-Seq sensitivity and throughput generated the capacity to profile whole transcriptomes of individual cells that have revolutionized the resolution and information of biological systems. The chapter discusses applications of these technologies to pathogen transcriptomics and how they advance our understanding of infection. During early infection, bacterial pathogens must sense and adaptively respond to the host environmental cues by altering their gene expression to enhance virulence, modulating metabolic pathways to utilize available resources, or evading host immune surveillance mechanisms. The current antibiotic resistance crisis necessitates the development of improved and accurate diagnostic methods that go beyond relying solely on growth rates and inhibition on selective media.
-
48.(2025) Cell Reports. 44, 5, 115609. Abstract[All authors]
Microglia are parenchymal brain macrophages that are established during embryogenesis and form a self-containing cellular compartment that resists seeding with cells derived from adult definitive hematopoiesis. We report that monocyte-derived macrophages (MoMΦs) accumulate in the brain of aging mice with distinct topologies, including the nigrostriatum and medulla but not the frontal cortex. Parenchymal MoMΦs adopt bona fide microglia morphology and expression profiles. Due to their hematopoietic stem cell (HSC) derivation, monocyte-derived microglia (MoMg) are unlike yolk-sac-derived cells, targets of clonal hematopoiesis (CH). Indeed, using a chimeric transfer model, we show that the hematopoietic expression of DNMT3AR882H, a prominent human CH variant, renders MoMg pathogenic and promotes motor deficits resembling atypical Parkinsonian disorders. Collectively, we establish that MoMg progressively seed the brain of healthy aging mice, accumulate in selected areas, and, when carrying a somatic mutation associated with CH, can cause brain pathology.
-
47.(2025) eLife. 13, RP100922. Abstract
Trained immunity (TI) is the process wherein innate immune cells gain functional memory upon exposure to specific ligands or pathogens, leading to augmented inflammatory responses and pathogen clearance upon secondary exposure. While the differentiation of hematopoietic stem cells (HSCs) and reprogramming of bone marrow (BM) progenitors are well-established mechanisms underpinning durable TI protection, remodeling of the cellular architecture within the tissue during TI remains underexplored. Here, we study the effects of peritoneal Bacillus Calmette-Guérin (BCG) administration to find TI-mediated protection in the spleen against a subsequent heterologous infection by the Gram-negative pathogen Salmonella Typhimurium (S.Tm). Utilizing single cell RNA-sequencing and flow cytometry, we discerned STAT1-regulated genes in TI-associated resident and recruited splenic myeloid populations. The temporal dynamics of TI were further elucidated, revealing both early and delayed myeloid subsets with time-dependent, cell-type-specific STAT1 signatures. Using lineage tracing, we find that tissue-resident red pulp macrophages (RPM), initially depleted by BCG exposure, are restored from both tissue-trained, self-renewing macrophages and from bone marrow-derived progenitors, fostering long lasting local defense. Early inhibition of STAT1 activation, using specific JAK-STAT inhibitors, reduces both RPM loss and recruitment of trained monocytes. Our study suggests a temporal window soon after BCG vaccination, in which STAT1-dependent activation of long-lived resident cells in the tissue mediates localized protection.
2024
-
46.(2024) STAR Protocols. 5, 3, 103137. Abstract
Ribosome quantification in single cells is typically achieved through fluorescence tagging of ribosomal proteins. Here, we present a protocol for comparing ribosomal levels in bacteria at different growth stages using fluorescence in situ hybridization of rRNA (rRNA-FISH), eliminating the need for genetic engineering of the strain of interest. We detail the steps for preparing bacterial samples, staining with fluorescent probes, and acquiring data using flow cytometry and microscopy. Furthermore, we provide guidelines on controlling for proper labeling through signal localization analysis. For complete details on the use and execution of this protocol, please refer to Ciolli Mattioli et al.1
-
45.
-
44.(2024) Fluorescence In Situ Hybridization (FISH). Haimovich G.(eds.). p. 3-23 Abstract
In this chapter, we describe in detail how to perform a successful smFISH experiment and how to quantify mRNA transcripts in bacterial cells. The flexibility of the method allows for straightforward adaptation to different bacterial species and experimental conditions. Thanks to the feasibility of the approach, the method can easily be adapted by other laboratories. Finally, we believe that this method has a great potential to generate insights into the complicated life of bacteria.
2023
-
43.(2023) Nature Communications. 14, 1, 2070. Abstract[All authors]
Both fatty bone marrow (FBM) and somatic mutations in hematopoietic stem cells (HSCs), also termed clonal hematopoiesis (CH) accumulate with human aging. However it remains unclear whether FBM can modify the evolution of CH. To address this question, we herein present the interaction between CH and FBM in two preclinical male mouse models: after sub-lethal irradiation or after castration. An adipogenesis inhibitor (PPARγ inhibitor) is used in both models as a control. A significant increase in self-renewal can be detected in both human and rodent DNMT3AMut-HSCs when exposed to FBM. DNMT3AMut-HSCs derived from older mice interacting with FBM have even higher self-renewal in comparison to DNMT3AMut-HSCs derived from younger mice. Single cell RNA-sequencing on rodent HSCs after exposing them to FBM reveal a 6-10 fold increase in DNMT3AMut-HSCs and an activated inflammatory signaling. Cytokine analysis of BM fluid and BM derived adipocytes grown in vitro demonstrates an increased IL-6 levels under FBM conditions. Anti-IL-6 neutralizing antibodies significantly reduce the selective advantage of DNMT3AMut-HSCs exposed to FBM. Overall, paracrine FBM inflammatory signals promote DNMT3A-driven clonal hematopoiesis, which can be inhibited by blocking the IL-6 pathway.
-
42.(2023) Current Biology. 33, 22, p. 4880-4892.e14 Abstract
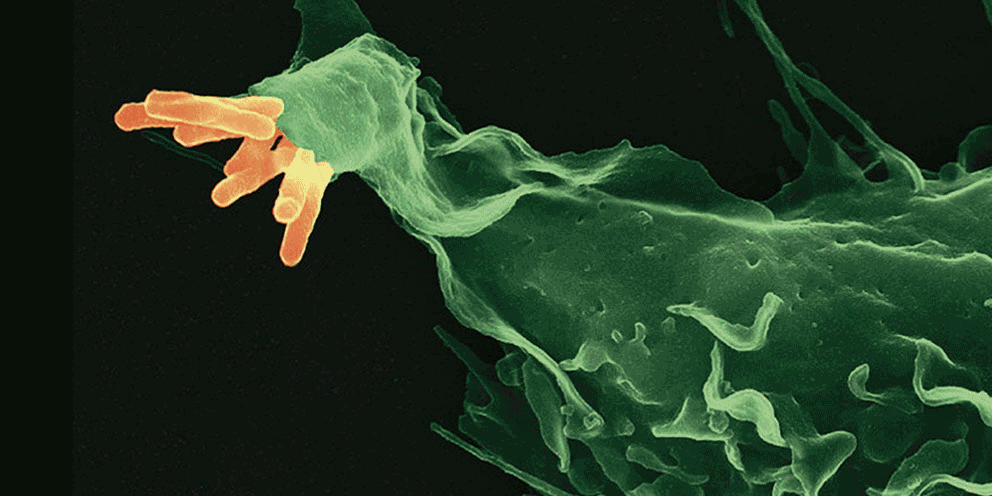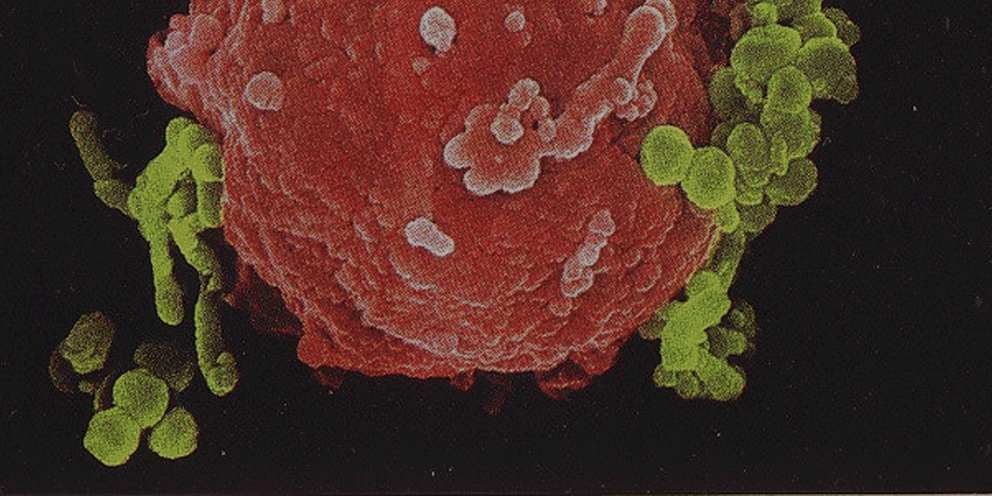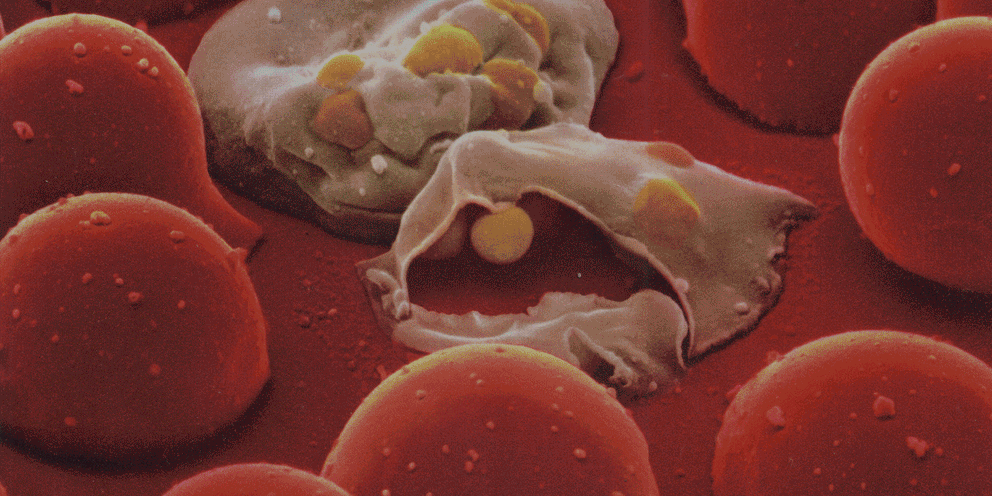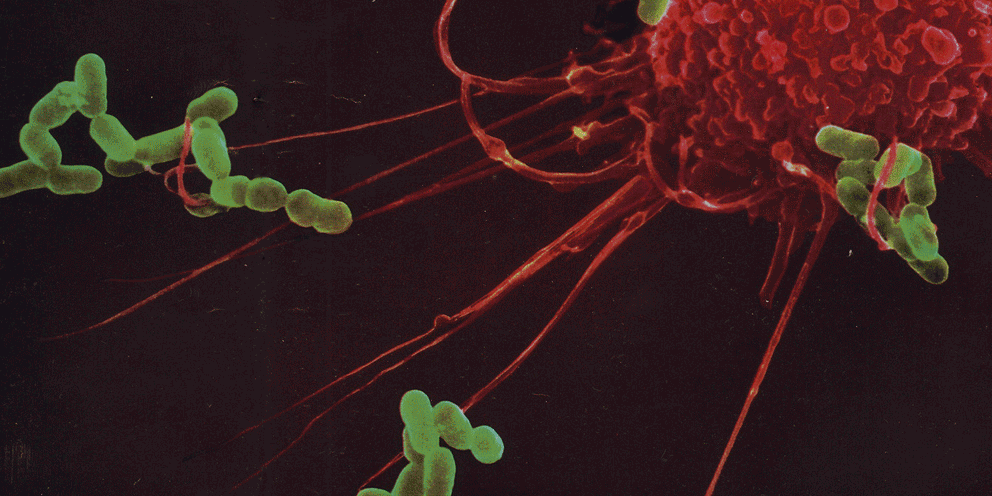
Bacteria undergo cycles of growth and starvation to which they must adapt swiftly. One important strategy for adjusting growth rates relies on ribosomal levels. Although high ribosomal levels are required for fast growth, their dynamics during starvation remain unclear. Here, we analyzed ribosomal RNA (rRNA) content of individual Salmonella cells by using fluorescence in situ hybridization (rRNA-FISH) and measured a dramatic decrease in rRNA numbers only in a subpopulation during nutrient limitation, resulting in a bimodal distribution of cells with high and low rRNA content. During nutritional upshifts, the two subpopulations were associated with distinct phenotypes. Using a transposon screen coupled with rRNA-FISH, we identified two mutants, DksA and RNase I, acting on rRNA transcription shutdown and degradation, which abolished the formation of the subpopulation with low rRNA content. Our work identifies a bacterial mechanism for regulation of ribosomal bimodality that may be beneficial for population survival during starvation.
-
41.(2023) Current Opinion in Immunology. 84, 102369. Abstract
Macrophages are phagocytic cells distributed across tissues that sustain homeostasis by constantly probing their local environment. Upon perturbations, macrophages rewire their energy metabolism to execute their immune programs. Intensive research in the field of immunometabolism highlights cell-intrinsic immunometabolites such as succinate and itaconate as immunomodulatory signals. A role for cell-extrinsic stimuli now emerges with evidence for signals that shape macrophages' metabolism in a tissue-specific manner. In this review, we will cover macrophage immunometabolism in the gut, a complex metabolic and immunologically active tissue. During homeostasis, gut macrophages are constantly exposed to pro-inflammatory ligands from the microbiota, and in contrast, are balanced by microbiota-derived anti-inflammatory metabolites. Given their extensive metabolic changes during activation, spatial analyses of the tissue will allow the characterization of metabolic niches of macrophage in the gut. Identifying metabolic perturbations of macrophage subsets during chronic inflammation and infection can direct future tissue-specific metabolotherapies.
-
40.(2023) Proceedings of the National Academy of Sciences. 120, 28, e221881212. Abstract
Encounters between host cells and intracellular bacterial pathogens lead to complex phenotypes that determine the outcome of infection. Single-cell RNA sequencing (scRNA-seq) is increasingly used to study the host factors underlying diverse cellular phenotypes but has limited capacity to analyze the role of bacterial factors. Here, we developed scPAIR-seq, a single-cell approach to analyze infection with a pooled library of multiplex-tagged, barcoded bacterial mutants. Infected host cells and barcodes of intracellular bacterial mutants are both captured by scRNA-seq to functionally analyze mutant-dependent changes in host transcriptomes. We applied scPAIR-seq to macrophages infected with a library of Salmonella Typhimurium secretion system effector mutants. We analyzed redundancy between effectors and mutant-specific unique fingerprints and mapped the global virulence network of each individual effector by its impact on host immune pathways. ScPAIR-seq is a powerful tool to untangle bacterial virulence strategies and their complex interplay with host defense strategies that drive infection outcome.
-
39.(2023) Cell. 186, 12, p. 2690-2704.e20 Abstract[All authors]
Biofilm formation is generally recognized as a bacterial defense mechanism against environmental threats, including antibiotics, bacteriophages, and leukocytes of the human immune system. Here, we show that for the human pathogen Vibrio cholerae, biofilm formation is not only a protective trait but also an aggressive trait to collectively predate different immune cells. We find that V. cholerae forms biofilms on the eukaryotic cell surface using an extracellular matrix comprising primarily mannose-sensitive hemagglutinin pili, toxin-coregulated pili, and the secreted colonization factor TcpF, which differs from the matrix composition of biofilms on other surfaces. These biofilms encase immune cells and establish a high local concentration of a secreted hemolysin to kill the immune cells before the biofilms disperse in a c-di-GMP-dependent manner. Together, these results uncover how bacteria employ biofilm formation as a multicellular strategy to invert the typical relationship between human immune cells as the hunters and bacteria as the hunted.
-
38.(2023) Infection and Immunity. 91, 4, Abstract
Bacterial pathogens can invade the tissue and establish a protected intracellular niche at the site of invasion that can spread locally (e.g., microcolonies) or to systemic sites (e.g., granulomas). Invasion of the tissue and establishment of intracellular infection are rare events that are difficult to study in the in vivo setting but have critical clinical consequences, such as long-term carriage, reinfections, and emergence of antibiotic resistance. Here, I discuss Salmonella interactions with its host macrophage during early stages of infection and their critical role in determining infection outcome. The dynamics of host-pathogen interactions entail highly heterogenous host immunity, bacterial virulence, and metabolic cross talk, requiring in vivo analysis at single-cell resolution. I discuss models and single-cell approaches that provide a global understanding of the establishment of a protected intracellular niche within the tissue and the host-pathogen landscape at infection bottlenecks during early stages of infection. Studying cellular host-pathogen interactions in vivo can improve our knowledge of the trajectory of infection between the initial inoculation with a dose of pathogens and the appearance of symptoms of disease.
-
37.(2023) eLife. 12, Abstract
Unicellular algae, termed phytoplankton, greatly impact the marine environment by serving as the basis of marine food webs and by playing central roles in the biogeochemical cycling of elements. The interactions between phytoplankton and heterotrophic bacteria affect the fitness of both partners. It is becoming increasingly recognized that metabolic exchange determines the nature of such interactions, but the underlying molecular mechanisms remain underexplored. Here, we investigated the molecular and metabolic basis for the bacterial lifestyle switch, from coexistence to pathogenicity, in Sulfitobacter D7 during its interaction with Emiliania huxleyi, a cosmopolitan bloom-forming phytoplankter. To unravel the bacterial lifestyle switch, we analyzed bacterial transcriptomes in response to exudates derived from algae in exponential growth and stationary phase, which supported the Sulfitobacter D7 coexistence and pathogenicity lifestyles, respectively. In pathogenic mode, Sulfitobacter D7 upregulated flagellar motility and diverse transport systems, presumably to maximize assimilation of E. huxleyi-derived metabolites released by algal cells upon cell death. Algal dimethylsulfoniopropionate (DMSP) was a pivotal signaling molecule that mediated the transition between the lifestyles, supporting our previous findings. However, the coexisting and pathogenic lifestyles were evident only in the presence of additional algal metabolites. Specifically, we discovered that algae-produced benzoate promoted the growth of Sulfitobacter D7 and hindered the DMSP-induced lifestyle switch to pathogenicity, demonstrating that benzoate is important for maintaining the coexistence of algae and bacteria. We propose that bacteria can sense the physiological state of the algal host through changes in the metabolic composition, which will determine the bacterial lifestyle during interaction.
2022
-
36.(2022) Nature. 606, 7914, p. 570-575 Abstract[All authors]
The lineage and developmental trajectory of a cell are key determinants of cellular identity. In the vascular system, endothelial cells (ECs) of blood and lymphatic vessels differentiate and specialize to cater to the unique physiological demands of each organ1,2. Although lymphatic vessels were shown to derive from multiple cellular origins, lymphatic ECs (LECs) are not known to generate other cell types3,4. Here we use recurrent imaging and lineage-tracing of ECs in zebrafish anal fins, from early development to adulthood, to uncover a mechanism of specialized blood vessel formation through the transdifferentiation of LECs. Moreover, we demonstrate that deriving anal-fin vessels from lymphatic versus blood ECs results in functional differences in the adult organism, uncovering a link between cell ontogeny and functionality. We further use single-cell RNA-sequencing analysis to characterize the different cellular populations and transition states involved in the transdifferentiation process. Finally, we show that, similar to normal development, the vasculature is rederived from lymphatics during anal-fin regeneration, demonstrating that LECs in adult fish retain both potency and plasticity for generating blood ECs. Overall, our research highlights an innate mechanism of blood vessel formation through LEC transdifferentiation, and provides in vivo evidence for a link between cell ontogeny and functionality in ECs.
-
35.(2022) Methods in Molecular Biology. p. 133-147 Abstract
Complex interactions between diverse host immune cells can determine the outcome of pathogen infections. Advances in single-cell RNA sequencing (scRNA-seq) allow detection of the transcriptional patterns of different immune cells at steady state and after infection. To reveal the complex interactions of the human immune system in response to diverse intracellular pathogens, we developed a protocol for scRNA-seq of ex vivo infected human peripheral blood mononuclear cells (PBMCs). We demonstrate here infection with Salmonella enterica serovar Typhimurium, but this protocol can be used for any other pathogen of interest, and expand our knowledge of human hostpathogen biology.
-
34.(2022) Nature Microbiology. 7, 4, p. 497-507 Abstract
Following detection of bacteria, macrophages switch their metabolism from oxidative respiration through the tricarboxylic acid cycle to high-rate aerobic glycolysis. This immunometabolic shift enables pro-inflammatory and antimicrobial responses and is facilitated by the accumulation of fatty acids, tricarboxylic acid-derived metabolites and catabolism of amino acids. Recent studies have shown that these immunometabolites are co-opted by pathogens as environmental cues for expression of virulence genes. We review mechanisms by which host immunometabolites regulate bacterial pathogenicity and discuss opportunities for the development of therapeutics targeting metabolic host-pathogen crosstalk.
-
33.(2022) Immunity. 55, 3, p. 442-458.e8 Abstract[All authors]
Consecutive exposures to different pathogens are highly prevalent and often alter the host immune response. However, it remains unknown how a secondary bacterial infection affects an ongoing adaptive immune response elicited against primary invading pathogens. We demonstrated that recruitment of Sca-1+ monocytes into lymphoid organs during Salmonella Typhimurium (STm) infection disrupted pre-existing germinal center (GC) reactions. GC responses induced by influenza, plasmodium, or commensals deteriorated following STm infection. GC disruption was independent of the direct bacterial interactions with B cells and instead was induced through recruitment of CCR2-dependent Sca-1+ monocytes into the lymphoid organs. GC collapse was associated with impaired cellular respiration and was dependent on TNFα and IFNγ, the latter of which was essential for Sca-1+ monocyte differentiation. Monocyte recruitment and GC disruption also occurred during LPS-supplemented vaccination and Listeria monocytogenes infection. Thus, systemic activation of the innate immune response upon severe bacterial infection is induced at the expense of antibody-mediated immunity.
2021
-
32.(2021) Immunity. 54, 12, p. 2712-2723.e6 Abstract
Interactions between intracellular bacteria and mononuclear phagocytes give rise to diverse cellular phenotypes that may determine the outcome of infection. Recent advances in single-cell RNA sequencing (scRNA-seq) have identified multiple subsets within the mononuclear population, but implications to their function during infection are limited. Here, we surveyed the mononuclear niche of intracellular Salmonella Typhimurium (S.Tm) during early systemic infection in mice. We described eclipse-like growth kinetics in the spleen, with a first phase of bacterial control mediated by tissue-resident red-pulp macrophages. A second phase involved extensive bacterial replication within a macrophage population characterized by CD9 expression. We demonstrated that CD9+ macrophages induced pathways for detoxificating oxidized lipids, that may be utilized by intracellular S.Tm. We established that CD9+ macrophages originated from non-classical monocytes (NCM), and NCM-depleted mice were more resistant to S.Tm infection. Our study defines macrophage subset-specific host-pathogen interactions that determine early infection dynamics and infection outcome of the entire organism.
-
31.(2021) Science. 371, 6527, p. 400-405 Abstract[All authors]
Key to the success of intracellular pathogens is the ability to sense and respond to a changing host cell environment. Macrophages exposed to microbial products undergo metabolic changes that drive inflammatory responses. However, the role of macrophage metabolic reprogramming in bacterial adaptation to the intracellular environment has not been explored. Here, using metabolic profiling and dual RNA sequencing, we show that succinate accumulation in macrophages is sensed by intracellular Salmonella Typhimurium (S. Tm) to promote antimicrobial resistance and type III secretion. S. Tm lacking the succinate uptake transporter DcuB displays impaired survival in macrophages and in mice. Thus, S. Tm co-opts the metabolic reprogramming of infected macrophages as a signal that induces its own virulence and survival, providing an additional perspective on metabolic host-pathogen cross-talk.
2019
-
30.(2019) Scientific Reports. 9, 1, 19244. Abstract
Dual transcriptional profiling of host and bacteria during infection is challenging due to the low abundance of bacterial mRNA. We report Pathogen Hybrid Capture (PatH-Cap), a method to enrich for bacterial mRNA and deplete bacterial rRNA simultaneously from dual RNA-seq libraries using transcriptome-specific probes. By addressing both the differential RNA content of the host relative to the infecting bacterium and the overwhelming abundance of uninformative structural RNAs (rRNA, tRNA) of both species in a single step, this approach enables analysis of very low-input RNA samples. By sequencing libraries before (pre-PatH-Cap) and after (post-PatH-Cap) enrichment, we achieve dual transcriptional profiling of host and bacteria, respectively, from the same sample. Importantly, enrichment preserves relative transcript abundance and increases the number of unique bacterial transcripts per gene in post-PatH-Cap libraries compared to pre-PatH-Cap libraries at the same sequencing depth, thereby decreasing the sequencing depth required to fully capture the transcriptional profile of the infecting bacteria. We demonstrate that PatH-Cap enables the study of low-input samples including single eukaryotic cells infected by 13 Pseudomonas aeruginosa bacteria and paired host-pathogen temporal gene expression analysis of Mycobacterium tuberculosis infecting macrophages. PatH-Cap can be applied to the study of a range of pathogens and microbial species, and more generally, to lowly-abundant species in mixed populations.
[All authors] -
29.(2019) Nature Communications. 10, 1, 3266. Abstract
Complex interactions between different host immune cell types can determine the outcome of pathogen infections. Advances in single cell RNA-sequencing (scRNA-seq) allow probing of these immune interactions, such as cell-type compositions, which are then interpreted by deconvolution algorithms using bulk RNA-seq measurements. However, not all aspects of immune surveillance are represented by current algorithms. Here, using scRNA-seq of human peripheral blood cells infected with Salmonella, we develop a deconvolution algorithm for inferring cell-type specific infection responses from bulk measurements. We apply our dynamic deconvolution algorithm to a cohort of healthy individuals challenged ex vivo with Salmonella, and to three cohorts of tuberculosis patients during different stages of disease. We reveal cell-type specific immune responses associated not only with ex vivo infection phenotype but also with clinical disease stage. We propose that our approach provides a predictive power to identify risk for disease, and human infection outcomes.
2018
-
28.(2018) Current Opinion in Microbiology. 42, p. 31-39 Abstract
Despite the availability of antibiotics and immunization, infectious diseases remain a major cause of malignancy and death worldwide. Yet, it is well documented that for most infectious agents, clinical disease develops in only a small minority of infected individuals. There is, in fact, great heterogeneity in infection outcome, from complete clearance of the pathogen to severe illness. Understanding this variation remains elusive, despite its great potential to equip us with new tools for the treatment of infectious diseases. Here, we propose a novel perspective for studying this diversity in human infection outcome, one that utilizes single-cell analysis technologies. Recent advances in single-cell RNA-seq technologies allow the detection of rare subpopulations that play important roles in host-pathogen interactions. We propose that applying single-cell RNA-seq to the study of infection can provide a 'fingerprint' of the immune cell types that are associated with the ability of the host to clear a pathogen and, thereby, broaden our current understanding of variation in susceptibility to infection within the population.
2017
-
27.(2017) Genome Biology. 18, 1, 200. Abstract
The interaction between a pathogen and a host is a highly dynamic process in which both agents activate complex programs. Here, we introduce a single-cell RNA-sequencing method, scDual-Seq, that simultaneously captures both host and pathogen transcriptomes. We use it to study the process of infection of individual mouse macrophages with the intracellular pathogen Salmonella typhimurium. Among the infected macrophages, we find three subpopulations and we show evidence for a linear progression through these subpopulations, supporting a model in which these three states correspond to consecutive stages of infection.
-
26.(2017) Science. 357, 6356, p. 1156-1160 Abstract[All authors]
Growing evidence suggests that microbes can influence the efficacy of cancer therapies. By studying colon cancer models, we found that bacteria can metabolize the chemotherapeutic drug gemcitabine (2,2-difluorodeoxycytidine) into its inactive form, 2,2-difluorodeoxyuridine. Metabolism was dependent on the expression of a long isoform of the bacterial enzyme cytidine deaminase (CDDL), seen primarily in Gammaproteobacteria. In a colon cancer mouse model, gemcitabine resistance was induced by intratumor Gammaproteobacteria, dependent on bacterial CDDL expression, and abrogated by cotreatment with the antibiotic ciprofloxacin. Gemcitabine is commonly used to treat pancreatic ductal adenocarcinoma (PDAC), and we hypothesized that intratumor bacteria might contribute to drug resistance of these tumors. Consistent with this possibility, we found that of the 113 human PDACs that were tested, 86 (76%) were positive for bacteria, mainly Gammaproteobacteria.
-
25.(2017) PLoS Pathogens. 13, 5, e1006363. Abstract[All authors]
A key to the pathogenic success of Mycobacterium tuberculosis (Mtb), the causative agent of tuberculosis, is the capacity to survive within host macrophages. Although several factors required for this survival have been identified, a comprehensive knowledge of such factors and how they work together to manipulate the host environment to benefit bacterial survival are not well understood. To systematically identify Mtb factors required for intracellular growth, we screened an arrayed, non-redundant Mtb transposon mutant library by high-content imaging to characterize the mutant-macrophage interaction. Based on a combination of imaging features, we identified mutants impaired for intracellular survival. We then characterized the phenotype of infection with each mutant by profiling the induced macrophage cytokine response. Taking a systems-level approach to understanding the biology of identified mutants, we performed a multiparametric analysis combining pathogen and host phenotypes to predict functional relationships between mutants based on clustering. Strikingly, mutants defective in two well-known virulence factors, the ESX-1 protein secretion system and the virulence lipid phthiocerol dimycocerosate (PDIM), clustered together. Building upon the shared phenotype of loss of the macrophage type I interferon (IFN) response to infection, we found that PDIM production and export are required for coordinated secretion of ESX-1-substrates, for phagosomal permeabilization, and for downstream induction of the type I IFN response. Multiparametric clustering also identified two novel genes that are required for PDIM production and induction of the type I IFN response. Thus, multiparametric analysis combining host and pathogen infection phenotypes can be used to identify novel functional relationships between genes that play a role in infection.
-
24.(2017) Current Opinion in Microbiology. 36, p. 69-75 Abstract
Most of our understanding of the hostbacterium interaction has come from studies of bulk populations. In reality, highly adaptable and dynamic host cells and bacteria engage in complex, diverse interactions. This complexity necessarily limits the depth of understanding that can be gained with bulk population measurements. Here, we will review the merit of single cell analysis to characterize this diversity that can trigger heterogeneous outcomes. We will discuss heterogeneity of bacterial and host populations, differences in host microenvironments, technological advances that facilitate the analysis of rare subpopulations, and the potential relevance of these subpopulations to infection outcomes. We focus our discussion on intracellular bacterial pathogens and on methods that characterize and quantify RNA in single cells, aiming to highlight how novel methodologies have the potential to characterize the multidimensional process of infection and to provide answers to some of the most fundamental questions in the field.
2016
-
23.(2016) Gut Microbes. 7, 6, p. 518-525 Abstract
The interface between immune cells and intracellular bacterial pathogens produces complex, diverse interactions. During individual encounters, highly adaptable and dynamic host cells and bacteria vary in their responses, thereby contributing to well-documented heterogeneous outcomes of infection. The challenge now is to break down the multidimensionality of these interactions into informative readouts of population physiology and predictors of responses to infection. We recently reported one approach to this challenge that couples single cell RNA-seq analysis with fluorescent markers to characterize infection phenotype.1 We detected bacterial subpopulations that elicit profoundly different host responses to infection in the specific host cells that they infect. Here we describe how heterogeneity might be maintained in populations of host and pathogens and discuss the advantages that heterogeneity confers to bacteria during infection and to host cells for eradicating a pathogen. We propose that single cell studies will allow the unraveling of host-pathogen biology and lead to an understanding of how the sum of individual encounters leads to the infection outcome of a whole organism.
-
22.(2016) Nature protocols. 11, 8, p. 1477-1491 Abstract
The ability to simultaneously characterize the bacterial and host expression programs during infection would facilitate a comprehensive understanding of pathogen-host interactions. Although RNA sequencing (RNA-seq) has greatly advanced our ability to study the transcriptomes of prokaryotes and eukaryotes separately, limitations in existing protocols for the generation and analysis of RNA-seq data have hindered simultaneous profiling of host and bacterial pathogen transcripts from the same sample. Here we provide a detailed protocol for simultaneous analysis of host and bacterial transcripts by RNA-seq. Importantly, this protocol details the steps required for efficient host and bacteria lysis, barcoding of samples, technical advances in sample preparation for low-yield sample inputs and a computational pipeline for analysis of both mammalian and microbial reads from mixed host-pathogen RNA-seq data. Sample preparation takes 3 d from cultured cells to pooled libraries. Data analysis takes an additional day. Compared with previous methods, the protocol detailed here provides a sensitive, facile and generalizable approach that is suitable for large-scale studies and will enable the field to obtain in-depth analysis of host-pathogen interactions in infection models.
2015
-
21.(2015) Cell. 162, 6, p. 1309-1321 Abstract[All authors]
Encounters between immune cells and invading bacteria ultimately determine the course of infection. These interactions are usually measured in populations of cells, masking cell-to-cell variation that may be important for infection outcome. To characterize the gene expression variation that underlies distinct infection outcomes and monitor infection phenotypes, we developed an experimental system that combines single-cell RNA-seq with fluorescent markers. Probing the responses of individual macrophages to invading Salmonella, we find that variation between individual infected host cells is determined by the heterogeneous activity of bacterial factors in individual infecting bacteria. We illustrate how variable PhoPQ activity in the population of invading bacteria drives variable host type I IFN responses by modifying LPS in a subset of bacteria. This work demonstrates a causative link between host and bacterial variability, with cell-to-cell variation between different bacteria being sufficient to drive radically different host immune responses. This co-variation has implications for host-pathogen dynamics in vivo.
2012
-
20.(2012) Proceedings of the National Academy of Sciences of the United States of America. 109, 46, p. 18839-18844 Abstract[All authors]
The search for developmental mechanisms driving vertebrate organogenesis has paved the way toward a deeper understanding of birth defects. During embryogenesis, parts of the heart and craniofacial muscles arise from pharyngeal mesoderm (PM) progenitors. Here, we reveal a hierarchical regulatory network of a set of transcription factors expressed in the PM that initiates heart and craniofacial organogenesis. Genetic perturbation of this network in mice resulted in heart and craniofacial muscle defects, revealing robust cross-regulation between its members. We identified Lhx2 as a previously undescribed player during cardiac and pharyngeal muscle development. Lhx2 and Tcf21 genetically interact with Tbx1, the major determinant in the etiology of DiGeorge/velo-cardio-facial/22q11.2 deletion syndrome. Furthermore, knockout of these genes in the mouse recapitulates specific cardiac features of this syndrome. We suggest that PM-derived cardiogenesis and myogenesis are network properties rather than properties specific to individual PM members. These findings shed new light on the developmental underpinnings of congenital defects.
-
19.(2012) Nucleic Acids Research. 40, 21, p. 10614-10627 Abstract
MicroRNAs (miRs) function primarily as post-transcriptional negative regulators of gene expression through binding to their mRNA targets. Reliable prediction of a miR's targets is a considerable bioinformatic challenge of great importance for inferring the miR's function. Sequence-based prediction algorithms have high false-positive rates, are not in agreement, and are not biological context specific. Here we introduce CoSMic (Context-Specific MicroRNA analysis), an algorithm that combines sequence-based prediction with miR and mRNA expression data. CoSMic differs from existing methods-it identifies miRs that play active roles in the specific biological system of interest and predicts with less false positives their functional targets. We applied CoSMic to search for miRs that regulate the migratory response of human mammary cells to epidermal growth factor (EGF) stimulation. Several such miRs, whose putative targets were significantly enriched by migration processes were identified. We tested three of these miRs experimentally, and showed that they indeed affected the migratory phenotype; we also tested three negative controls. In comparison to other algorithms CoSMic indeed filters out false positives and allows improved identification of context-specific targets. CoSMic can greatly facilitate miR research in general and, in particular, advance our understanding of individual miRs' function in a specific context.
-
18.(2012) EMBO Molecular Medicine. 4, 11, p. 1214-1229 Abstract[All authors]
Deregulated proliferation is a hallmark of cancer cells. Here, we show that microRNA-10b* is a master regulator of breast cancer cell proliferation and is downregulated in tumoural samples versus matched peritumoural counterparts. Two canonical CpG islands (5kb) upstream from the precursor sequence are hypermethylated in the analysed breast cancer tissues. Ectopic delivery of synthetic microRNA-10b* in breast cancer cell lines or into xenograft mouse breast tumours inhibits cell proliferation and impairs tumour growth in vivo, respectively. We identified and validated in vitro and in vivo three novel target mRNAs of miR-10b* (BUB1, PLK1 and CCNA2), which play a remarkable role in cell cycle regulation and whose high expression in breast cancer patients is associated with reduced disease-free survival, relapse-free survival and metastasis-free survival when compared to patients with low expression. This also suggests that restoration of microRNA-10b* expression might have therapeutic promise.
-
17.(2012) Molecular Cancer. 11, 44. Abstract
Background: Metastatic melanoma is a devastating disease with limited therapeutic options. MicroRNAs (miRNAs) are small non coding RNA molecules with important roles in post-transcriptional gene expression regulation, whose aberrant expression has been implicated in cancer.Results: We show that the expression of miRNAs from a large cluster on human chromosome 14q32 is significantly down-regulated in melanoma cell lines, benign nevi and melanoma samples relative to normal melanocytes. This miRNA cluster resides within a parentally imprinted chromosomal region known to be important in development and differentiation. In some melanoma cell lines, a chromosomal deletion or loss-of-heterozygosity was observed in the cis-acting regulatory region of this cluster. In several cell lines we were able to re-express two maternally-induced genes and several miRNAs from the cluster with a combination of de-methylating agents and histone de-acetylase inhibitors, suggesting that epigenetic modifications take part in their silencing. Stable over-expression of mir-376a and mir-376c, two miRNAs from this cluster that could be re-expressed following epigenetic manipulation, led to modest growth retardation and to a significant decrease in migration in-vitro. Bioinformatic analysis predicted that both miRNAs could potentially target the 3'UTR of IGF1R. Indeed, stable expression of mir-376a and mir-376c in melanoma cells led to a decrease in IGF1R mRNA and protein, and a luciferase reporter assay indicated that the 3'UTR of IGF1R is a target of both mir-376a and mir-376c.Conclusions: Our work is the first to show that the large miRNA cluster on chromosome 14q32 is silenced in melanoma. Our results suggest that down-regulation of mir-376a and mir-376c may contribute to IGF1R over-expression and to aberrant negative regulation of this signaling pathway in melanoma, thus promoting tumorigenesis and metastasis.
-
16.(2012) FASEB Journal. 26, 4, p. 1582-1592 Abstract[All authors]
The signaling pathways that commit cells to migration are incompletely understood. We employed human mammary cells and two stimuli: epidermal growth factor (EGF), which induced cellular migration, and serum factors, which stimulated cell growth. In addition to strong activation of ERK by EGF, and AKT by serum, early transcription remarkably differed: while EGF induced early growth response-1 (EGR1), and this was required for migration, serum induced c-Fos and FosB to enhance proliferation. We demonstrate that induction of EGR1 involves ERK-mediated down-regulation of microRNA-191 and phosphorylation of the ETS2 repressor factor (ERF) repressor, which subsequently leaves the nucleus. Unexpectedly, knockdown of ERF inhibited migration, which implies migratory roles for exported ERF molecules. On the other hand, chromatin immunoprecipitation identified a subset of direct EGR1 targets, including EGR1 autostimulation and SERPINB2, whose transcription is essential for EGF-induced cell migration. In summary, EGR1 and the EGF-ERK-ERF axis emerge from our study as major drivers of growth factor-induced mammary cell migration.
-
15.(2012) Journal of Mammary Gland Biology and Neoplasia. 17, 1, p. 3-14 Abstract
Signaling networks are involved in development, as well as in malignancy of the mammary gland. Distinct external stimuli activate intricate signaling cascades, which culminate in the activation of specific transcriptional programs. These signal-specific transcriptional programs are instigated by transcription factors (TFs) encoded by the immediate early genes (IEGs), and they lead to diverse cellular outcomes, including oncogenesis. Hence, regulating the expression of IEGs is of great importance, and involves several complementary transcriptional and posttranscriptional mechanisms, the latter entails also microRNAs (miRNAs). miRNAs are a class of non-coding RNAs, which have been implicated in regulation of various aspects of signaling networks. Through examination of the basic characteristics of miRNA function, we highlight the benefits of using miRNAs as regulators of early TFs and signaling networks. We further focus on the role of miRNAs as regulators of IEGs, which shape the initial steps of signaling-induced transcription. We especially emphasize the role of miRNAs in buffering external noise and maintaining low basal activation of IEGs in the absence of proper stimuli.
-
14.(2012) Biochemical Society Transactions. 40, 1, p. 26-30 Abstract
Stringent regulation of biochemical signalling pathways involves feedback and feedforward loops, which underlie robust cellular responses to external stimuli. Regulation occurs in all horizontal layers of signalling networks, primarily by proteins that mediate internalization of receptor-ligand complexes, dephosphorylation of kinases and their substrates, as well as transcriptional repression. Recent studies have unveiled the role of miRNAs (microRNAs), post-transcriptional regulators that control mRNA stability, as key modulators of signal propagation. By acting as genetic switches or fine-tuners, miRNAs can directly and multiply regulate cellular outcomes in response to diverse extracellular signals. Conversely, signalling networks temporally control stability, biogenesis and abundance of miRNAs, by regulating layers of the miRNA biogenesis pathway. In the present mini-review, we use a set of examples to illustrate the extensive interdependence between miRNAs and signalling networks.
2011
-
13.(2011) Molecular Cell. 42, 4, p. 524-535 Abstract[All authors]
Normal cells require continuous exposure to growth factors in order to cross a restriction point and commit to cell-cycle progression. This can be replaced by two short, appropriately spaced pulses of growth factors, where the first pulse primes a process, which is completed by the second pulse, and enables restriction point crossing. Through integration of comprehensive proteomic and transcriptomic analyses of each pulse, we identified three processes that regulate restriction point crossing: (1) The first pulse induces essential metabolic enzymes and activates p53-dependent restraining processes. (2) The second pulse eliminates, via the PI3K/AKT pathway, the suppressive action of p53, as well as (3) sets an ERK-EGR1 threshold mechanism, which digitizes graded external signals into an all-or-none decision obligatory for S phase entry. Together, our findings uncover two gating mechanisms, which ensure that cells ignore fortuitous growth factors and undergo proliferation only in response to consistent mitogenic signals.
-
12.(2011) Nature Reviews Molecular Cell Biology. 12, 2, p. 104-117 Abstract
Human-made information relay systems invariably incorporate central regulatory components, which are mirrored in biological systems by dense feedback and feedforward loops. This type of system control is exemplified by positive and negative feedback loops (for example, receptor endocytosis and dephosphorylation) that enable growth factors and receptor Tyr kinases of the epidermal growth factor receptor (EGFR)/ERBB family to regulate cellular function. Recent studies show that the collection of feedback regulatory loops can perform computational tasks - such as decoding ligand specificity, transforming graded input signals into a digital output and regulating response kinetics. Aberrant signal processing and feedback regulation can lead to defects associated with pathologies such as cancer.
-
11.(2011) PLoS ONE. 6, 4, e19246. Abstract
In leukemia patients, stress and anxiety were suggested to predict poorer prognosis. Oncological patients experience ample physiological and psychological stress, potentially leading to increased secretion of stress factors, including epinephrine, corticosteroids, and prostaglandins. Here we tested whether environmental stress and these stress factors impact survival of leukemia-challenged rats, and studied mediating mechanisms. F344 rats were administered with a miniscule dose of 60 CRNK-16 leukemia cells, and were subjected to intermittent forced swim stress or to administration of physiologically relevant doses of epinephrine, prostaglandin-E2 or corticosterone. Stress and each stress factor, and/or their combinations, doubled mortality rates when acutely applied simultaneously with, or two or six days after tumor challenge. Acute administration of the β-adrenergic blocker nadolol diminished the effects of environmental stress, without affecting baseline survival rates. Prolonged β-adrenergic blockade or COX inhibition (using etodolac) also increased baseline survival rates, possibly by blocking tumor-related or normal levels of catecholamines and prostaglandins. Searching for mediating mechanisms, we found that each of the stress factors transiently suppressed NK activity against CRNK-16 and YAC-1 lines on a per NK basis. In contrast, the direct effects of stress factors on CRNK-16 proliferation, vitality, and VEGF secretion could not explain or even contradicted the in vivo survival findings. Overall, it seems that environmental stress, epinephrine, and prostaglandins promote leukemia progression in rats, potentially through suppressing cell mediated immunity. Thus, patients with hematological malignancies, which often exhibit diminished NK activity, may benefit from extended β-blockade and COX inhibition.
2010
-
10.(2010) Brain, Behavior, and Immunity. 24, 6, p. 952-958 Abstract
Background: A unique opportunity to eradicate cancer is presented immediately after the excision of the primary tumor, but surgical procedures often induce the release of immunosuppressing factors that render cell mediated immunity ineffective. Here we tested the hypothesis that integration of peri-operative immunostimulation and blockade of immunosuppression could synergistically improve post-operative anti-metastatic immunity and long-term survival. Methods: Two syngeneic tumor models in F344 rats were employed, studying post-operative tumor progression. In the first model, survival following laparotomy and CRNK-16 leukemia was studied. Rats were peri-operatively treated with the immuno-stimulant poly I-C (5 × 0.2. mg/kg/inj), with catecholamine- and prostaglandin-blockers (shown to prevent post-operative immunosuppression: 4.5. mg/kg nadolol, 4. mg/kg indomethacin), with both interventions, or with neither. Long-term survival was assessed thereafter. The second model used the MADB106 mammary adenocarcinoma, assessing its lung tumor retention (LTR) following i.v. inoculation, as well as host marginating-pulmonary NK numbers and activity against this tumor. IL-12 was employed for immunostimulation (4 × 1.5 μg/kg/inj), with and without the above blockers. Results: Post-operative CRNK-16 survival rates were significantly improved only by the integrated approach of immune stimulation and endocrine blockers. Post-operative MADB106 LTR was additively reduced by the two interventions. Importantly, while IL-12 increased pulmonary NK cytotoxicity against MADB106, surgery markedly suppressed this cytotoxicity in both IL-12 and vehicle treated animals. The blockers prevented this suppression per lung and per single NK cell. Conclusions: Immunostimulation could be rendered ineffective post-operatively due to immunosuppression; therefore integrating endocrine-blocker therapies into the realm of peri-operative immunotherapy could optimize immune control over residual disease, potentially improving clinical outcomes.
-
9.(2010) Science Signaling. 3, 124, p. ra43 Abstract[All authors]
Epidermal growth factor (EGF) stimulates cells by launching gene expression programs that are frequently deregulated in cancer. MicroRNAs, which attenuate gene expression by binding complementary regions in messenger RNAs, are broadly implicated in cancer. Using genome-wide approaches, we showed that EGF stimulation initiates a coordinated transcriptional program of microRNAs and transcription factors. The earliest event involved a decrease in the abundance of a subset of 23 microRNAs. This step permitted rapid induction of oncogenic transcription factors, such as c-FOS, encoded by immediate early genes. In line with roles as suppressors of EGF receptor (EGFR) signaling, we report that the abundance of this early subset of microRNAs is decreased in breast and in brain tumors driven by the EGFR or the closely related HER2. These findings identify specific microRNAs as attenuators of growth factor signaling and oncogenesis.
-
8.(2010) Journal of Immunology. 184, 5, p. 2449-2457 Abstract
Clinical practice does not consider perioperative paracrine and neuroendocrine stress responses as risk factors for cancer recurrence, although recent animal studies provided supportive evidence. Suggested mechanisms include the effects of stress-hormones on tumor cells and on host physiology. In this study, in mice undergoing primary tumor excision, we tested the survival-enhancing potential of perioperative blockade of catecholamines and prostaglandins, and studied potential mediating mechanisms. C57BL/6J mice were inoculated intrafootpad with syngeneic B16F10.9-melanoma or Lewis lung carcinoma, and the paw was amputatedwhen a developing tumor exceeded 100 μl. The clinically used β-adrenergic antagonist propranolol, and/or the cyclooxygenase-2 inhibitor etodolac, were administered once before amputation, and recurrence-free survival was monitored. In different studies, NK cytotoxicity, leukocytes' molecular functional markers, and vascular endothelial growth factor secretion by tumor cells were studied in the context of surgery and drug treatments. The findings indicated that the combination of propranolol and etodolac, but neither drug alone, significantly and markedly improved survival rates in both tumor models, and was as effective as established immunostimulatory agents (IL-12 and polyinosinic-polycytiylic acid). Surgery markedly reduced NK cytotoxicity and NK cell expression of Fas ligand and CD11a, reduced all circulating lymphocyte-subtype concentrations, and increased corticosterone levels. Propranolol and etodolac administration counteracted these perturbations. B16 and 3LL secreted vascular endothelial growth factor in vitro, but secretion was not affected by catecholamine agonists, prostaglandins, corticosterone, propranolol, or etodolac. Overall, propranolol and etodolac administration, which could be applied perioperatively in most cancer patients with minimal risk and low cost, has counteracted several immunologic and endocrinologic perturbations and improved recurrence-free survival rates in mice undergoing primary tumor excision.
2008
-
7.(2008) Anesthesiology. 109, 6, p. 989-997 Abstract
BACKGROUND:: In cancer patients, allogeneic blood transfusion is associated with poorer prognosis, but the independent effect of the transfusion is controversial. Moreover, mediating mechanisms underlying the alleged cancer-promoting effects of blood transfusion are unknown, including the involvement of donors' leukocytes, erythrocytes, and soluble factors. METHOD:: Two syngeneic tumor models were used in Fischer 344 rats, the MADB106 mammary adenocarcinoma and the CRNK-16 leukemia. Outcomes included host ability to clear circulating cancer cells, and host survival rates. The independent impact of blood transfusion was assessed, and potential deleterious characteristics of the transfusion were studied, including blood storage duration; the role of erythrocytes, leukocyte, and soluble factors; and the kinetics of the effects. RESULTS:: Blood transfusion was found to be an independent and significant risk factor for cancer progression in both models, causing up to a fourfold increase in lung tumor retention and doubling mortality rates. Blood storage time was the critical determinant of these deleterious effects, regardless of whether the transfused blood was allogeneic or autogenic. Surprisingly, aged erythrocytes (9 days and older), rather than leukocytes or soluble factors, mediated the effects, which occurred in both operated and nonoperated animals. The effects of erythrocytes transfusion in the MADB106 model emerged immediately and dissipated within 24 h. CONCLUSIONS:: In rats, transfusion of fresh blood is less harmful than transfusion of stored blood in the context of progressing malignancies. Further studies should address mediating mechanisms through which erythrocytes'e storage duration can impact the rate of complications while treating malignant diseases and potentially other pathologies.
-
6.(2008) Annals of Surgical Oncology. 15, 7, p. 2042-2052 Abstract
Background: COX inhibitors and β-blockers were recently suggested to reduce cancer progression through inhibition of tumor proliferation and growth factor secretion, induction of tumor apoptosis, and prevention of cellular immune suppression during the critical perioperative period. Here we evaluated the perioperative impact of clinically applicable drugs from these categories in the context of surgery, studying natural killer (NK) cell activity and resistance to experimental metastases. Methods: F344 rats were treated with COX-1 inhibitors (SC560), COX-2 inhibitors (indomethacin, etodolac, or celecoxib), a β-blocker (propranolol), or a combination of a COX-2 inhibitor and a β-blocker (etodolac and propranolol). Rats underwent laparotomy, and were inoculated intravenously with syngeneic MADB106 tumor cells for the assessment of lung tumor retention (LTR). Additionally, the impact of these drug regimens on postoperative levels of NK cytotoxicity was studied in peripheral blood and marginating-pulmonary leukocytes. Results: Surgery increased MADB106 LTR. COX-2 inhibition, but not COX-1 inhibition, reduced postoperative LTR. Etodolac and propranolol both attenuated the deleterious impact of surgery, and their combined use abolished it. Surgery decreased NK cytotoxicity per NK cell in both immune compartments, and only the combination of etodolac and propranolol significantly attenuated these effects. Lastly, the initiation of drug treatment three days prior to surgery yielded the same beneficial effects as a single pre-operative administration, but, as discussed, prolonged treatment may be more advantageous clinically. Conclusions: Excess prostaglandin and catecholamine release contributes to postoperative immune-suppression. Treatment combining perioperative COX-2 inhibition and β-blockade is practical in operated cancer patients, and our study suggests potential immunological and clinical benefits.
-
5.(2008) Breast Cancer Research and Treatment. 107, 2, p. 211-223 Abstract
Despite a promising potential, interleukin-12 immunotherapy has yielded limited clinical success while causing perilous toxicities. Here we study a context in which IL-12 may prove clinically beneficial-the removal of the primary tumor, when cell-mediated immunity (CMI) may eradicate minimal residual disease (MRD), but is inhibited by postoperative immunosuppression, potentially leading to enhanced malignant progression. F344 rats were preoperatively treated with IL-12 and inoculated postoperatively with syngeneic MADB106 tumor cells. An optimal regimen of eight-day sustained exposure to IL-12 was developed (1 μg/rat/day), which caused mild side effects, increased baseline resistance to experimental MADB106 metastasis, and abolished the promotion of metastasis by laparotomy and other immunosuppressive paradigms. Depletion of NK cells indicated their major role in controlling MADB106 metastasis in naïve and IL-12 treated rats. Studying NK cytotoxicity, we found that IL-12 did not potentiate activity per NK cell, nor protected it from suppression by surgery. However, IL-12 increased the numbers of NK cells in the circulation and marginating pulmonary pool of naïve and operated rats, and correspondingly increased total NK activity in these compartments. Therefore, this study indicates anti-tumor effects of IL-12 based on increased numbers of strategically located NK cells, and advocates a prophylactic approach against the potential metastasis-promoting effects of surgery.
2007
-
4.(2007) Nature Cell Biology. 9, 8, p. 961-969 Abstract[All authors]
Cell migration driven by the epidermal growth factor receptor (EGFR) propels morphogenesis and involves reorganization of the actin cytoskeleton. Although de novo transcription precedes migration, transcript identity remains largely unknown. Through their actin-binding domains, tensins link the cytoskeleton to integrin-based adhesion sites. Here we report that EGF downregulates tensin-3 expression, and concomitantly upregulates cten, a tensin family member that lacks the actin-binding domain. Knockdown of cten or tensin-3, respectively, impairs or enhances mammary cell migration. Furthermore, cten displaces tensin-3 from the cytoplasmic tail of integrin β1, thereby instigating actin fibre disassembly. In invasive breast cancer, cten expression correlates not only with high EGFR and HER2, but also with metastasis to lymph nodes. Moreover, treatment of inflammatory breast cancer patients with an EGFR/HER2 dual-specificity kinase inhibitor significantly downregulated cten expression. In conclusion, a transcriptional tensin-3-cten switch may contribute to the metastasis of mammary cancer.
-
3.(2007) Brain Behavior And Immunity. 21, 4, p. 503-513 Abstract
Surgery may render patients susceptible to life-threatening complications, including infections and later metastases. Suppression of cell mediated immunity (CMI) and perturbations in the cytokine network were implicated in these outcomes. The current study assessed the effects of various surgeries on a wide array of immune indices, and compared patients' pre-operative immune status to that of control subjects. A total of 81 subjects (controls, moderate and major surgeries) provided up to five daily blood samples. Whole blood procedures were conducted within hours of blood withdrawal, assessing NK cell number and cytotoxicity, and plasma cytokine levels and induced production (IFNγ, IL-6, IL-10, and IL-12). Our findings indicate that surgery reduced NK cell numbers/ml blood, and independently suppressed NK activity per NK cell and per ml blood. Among other perturbations in the cytokine network, pro-CMI cytokine production (IL-12 and IFNγ) was reduced by surgery. Surprisingly, plasma levels of IFNγ and IL-6 increased following surgery, while their in vitro induced production showed opposite effects. Patients awaiting surgery exhibited impaired IL-12 induced production and NK activity/ml, and reduced IFNγ plasma levels. No significant associations were found between NK cytotoxicity and Th1 cytokines, although these indices showed high correlations with other variables. Overall, our findings indicate that patients exhibit impaired immune functions even before operation, which seem to contribute to the evident post-operative immune suppression. In the peri-operative context, induced cytokine production and plasma cytokines levels reflect different processes. Last, we suggest that peri-operative suppression of NK activity is mediated by neuroendocrine responses rather than Th1 cytokines.
-
2.
2006
-
1.(2006) Cancer Immunology, Immunotherapy. 55, 11, p. 1348-1357 Abstract
It is unclear whether autologous immunity could be recruited to restrict the progression of leukemia. Patients harboring leukemia commonly display suppressed cell mediated immunity, which may contribute to their inability to control the disease. Immune response against leukemia is evident in allogeneic HLA-mismatched bone marrow transplantation, implicating the involvement of NK cells. This graft-versus-leukemia (GVL) activity suggests that, if not suppressed, an autologous NK cell response could potentially control acute leukemia that had down-regulated HLA expression. In the current study we assessed the role of non-suppressed autologous NK cells in controlling a syngeneic highly malignant leukemia, the CRNK-16 line, that constitute a major cause of natural death in aged F344 rats. A minuscule dose of 60 CRNK-16 leukemia cells per rat was sufficient to induce 50% mortality rates, and animals that survived this challenge did not show improved survival upon a second challenge. The CRNK-16 line was found to exhibit low levels of MHC-I, and selective in vivo depletion of NK cells nullified in vitro NK activity against the CRNK-16 line and reduced survival rates from this leukemia. In vivo activation of NK cells, employing low doses of poly I-C or IL-12, increased in vitro NK activity against the leukemia and dramatically improved survival rates when treatment was initiated before, but not after leukemia inoculation. These results indicate the ability of competent autologous NK cells to restrict highly malignant non-immunogenic leukemia. Thereby, this model presents an opportunity to study specific in vivo NK-leukemia interactions.