In the very near future, our experimental work will be terminated (Retirement).
In the past, we investigated membrane protein biogenesis, structure, and function.
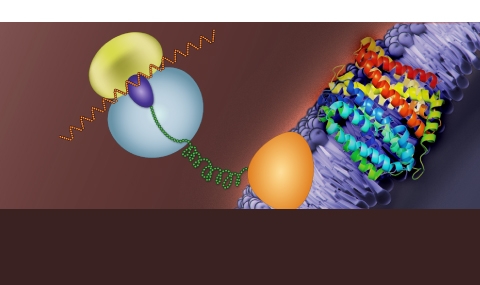
Biosynthetically, structurally and functionally, membrane proteins must follow interesting, dedicated principles. For example, unlike soluble proteins, most membrane proteins are translated by membrane-bound ribosomes and then assemble and function inside the lipophilic environment of the membrane.
Specifically, we asked how cells produce membrane proteins and how various structural determinants affect their function: (i) How ribosomes and mRNAs target the membrane, where localized translation of membrane proteins occurs. (ii) What dictates the fascinating capabilities of multidrug transporters and the multidrug-efflux phenomenon.