Dr. Liran Shlush
What triggers leukemia?
New scientists

When acute myeloid leukemia is first diagnosed, it appears as an overwhelming disease in need of immediate and aggressive treatment.
However, both physicians and scientists ignore the fact that the evolution of the disease is latent in the patient, and—in many cases—takes many years to develop. Dr. Liran Shlush believes that this long, latent period might be an opportunity for early treatment - but that it also hides great mysteries.
Dr. Shlush is a medical doctor who practices at Rambam Hospital in Haifa and joined the Weizmann Institute’s Department of Immunology in the fall. He has been investigating acute myeloid leukemia (AML) using a “cell genealogy” approach, which has led to key insights. AML is a cancer of the myeloid line of blood cells that is characterized by runaway growth of abnormal white blood cells accumulating in the bone marrow and interfering with the production of normal blood cells. Although it is a relatively rare disease - accounting for just 1.2 percent of cancer deaths in the U.S. - its incidence is expected to increase as the population ages.
By tracking the “family history” of individual stem cells from the blood of AML patients and using deep sequencing to identify alterations in genes commonly mutated in the disease, Dr. Shlush and his collaborators were able to identify - for the first time - pre-leukemic stem cells. These mutant stem cells go on to form cancerous cells.
Most cases of AML are diagnosed without any prior indication of cancer. Yet AML, like all cancers, arises from the multi-step accumulation of mutations. Dr. Shlush says that, on average, AML cells already carry about 13 mutations at the time of diagnosis. When AML patients are treated with chemotherapy, the cancerous cells are killed, but these mutant stem cells remain. This finding holds significant promise for treatment, earlier diagnosis, and new screening methods for AML and perhaps other cancers as well. The editors of Nature Medicine selected his paper on this finding as one of the most notable advances in medicine in 2014 with great clinical and scientific implications.
A fusion of medicine and research
Ever since his undergraduate years, Dr. Shlush, who is from Haifa—and today is married with three kids—has been fully focused on medicine. Over the years, he was increasingly drawn to basic research and says he understood that the fluid combination of the two offers the best promise for advancing human health. He completed his BSc in medical sciences with honors, received his medical degree from the Technion, and went on to serve as Deputy Director of the Israel Navy Medical Institute during his military service. He completed a PhD in population genetics in 2012 at the Technion, and did a three-year residency in internal medicine at Rambam.
In 2012, he packed his bags for the University of Toronto, where he spent the next few years as both a postdoctoral research fellow and a clinical fellow with a focus on leukemia at Princess Margaret Cancer Centre.
During his PhD and medical studies, Dr. Shlush studied leukemia evolution from diagnosis to relapse. He compared paired samples from AML patients at various stages of the disease and treatment and found that blood samples from AML patients at diagnosis contained the ancestral stem and progenitor cells that carried the initiating genetic events in AML. He was able to show that these leukemic stem cells actually survived chemotherapy and contributed to leukemia relapse. These studies have important implications for future approaches to prevent relapse by targeting the surviving pre-leukemic stem cells.
He recently initiated a clinical study to track large cohorts of AML patients by taking samples from diagnosis, after therapy induction, and every three months during remission until relapse. Dr. Shlush continues to collaborate with his postdoctoral mentor, Dr. John E. Dick, and clinical mentor, Dr. Mark Minden, at Princess Margaret to study AML and other blood malignancies.
At the Weizmann Institute, he is working with Prof. Amos Tanay from the Departments of Biological Regulation; and Computer Science and Applied Mathematics and Dr. Ido Amit from the Department of Immunology to look at the genetic and epigenetic regulation networks involved in these diseases.
Meanwhile, he continues to work as a hematologist at Rambam to follow leukemia and patients with preleukemic conditions. “My clinical work focuses on the treatment of individuals with a pre-leukemic condition called myelodysplastic syndrome,” he says. “In this disease, bone marrow failure is the end result of malignant growth; about 20 percent of these cases will progress to AML.”
Dr. Liran Shlush is supported by the Abramson Family Center for Young Scientists, ACWIS Midwest Gala donors, the Estate of Leah Arbel, the Benoziyo Fund for the Advancement of Science, CNRS-Centre National de la Recherche Scientifique, David and Fela Shapell Family Foundation INCPM Fund for Preclinical Studies, Estate of David Turner, and the Morris & Ruth Wagner Marek Sutkiewicz Laboratory for Cancer Research. He is the incumbent of the Ruth and Louis Leland Career Development Chair.
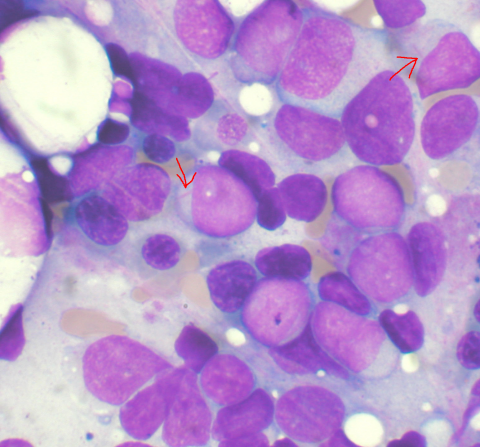
Acute myeloid leukemia cells in bone marrow