From Vascular Disorders to Treatment Discovery
We use zebrafish, patient-derived samples, and in vitro vascular models to uncover how genetic mutations and dysregulated signaling pathways lead to abnormal vessel growth and vascular dysfunction. Our work bridges fundamental mechanisms with disease biology, including the development of the first zebrafish model of Kaposiform Lymphangiomatosis (KLA) and the identification of pathways potentially implicated in cerebral small vessel disease. By integrating genetic data from patients with functional testing in living models, we aim to identify therapeutic targets and develop new strategies to treat vascular and lymphatic disorders.
The zebrafish (Danio rerio) is the second most widely used animal model in biomedical research and is essential for translational science due to its strong genetic similarity to humans, sharing orthologues for 71% of human genes and over 80% of known disease-associated genes. This genetic proximity, together with transparent embryos and rapid development outside the body, enables non-invasive, real-time observation of complex biological processes at a scale not possible in most mammalian models. Zebrafish are ideally suited for large-scale genetic manipulation, CRISPR-based genome editing, and high-throughput drug screening, allowing researchers to uncover disease mechanisms and test therapies quickly and efficiently. Discoveries made in zebrafish have already advanced research in genetic diseases, cancer, heart and developmental disorders, and have directly contributed to human clinical trials, making this small fish a powerful and ethically responsible bridge from laboratory discovery to life-saving patient treatments.
In our lab, we bring together leading minds to develop new therapies for rare human developmental disorders, using zebrafish-based drug screening to help thousands of people worldwide who currently lack effective treatments.
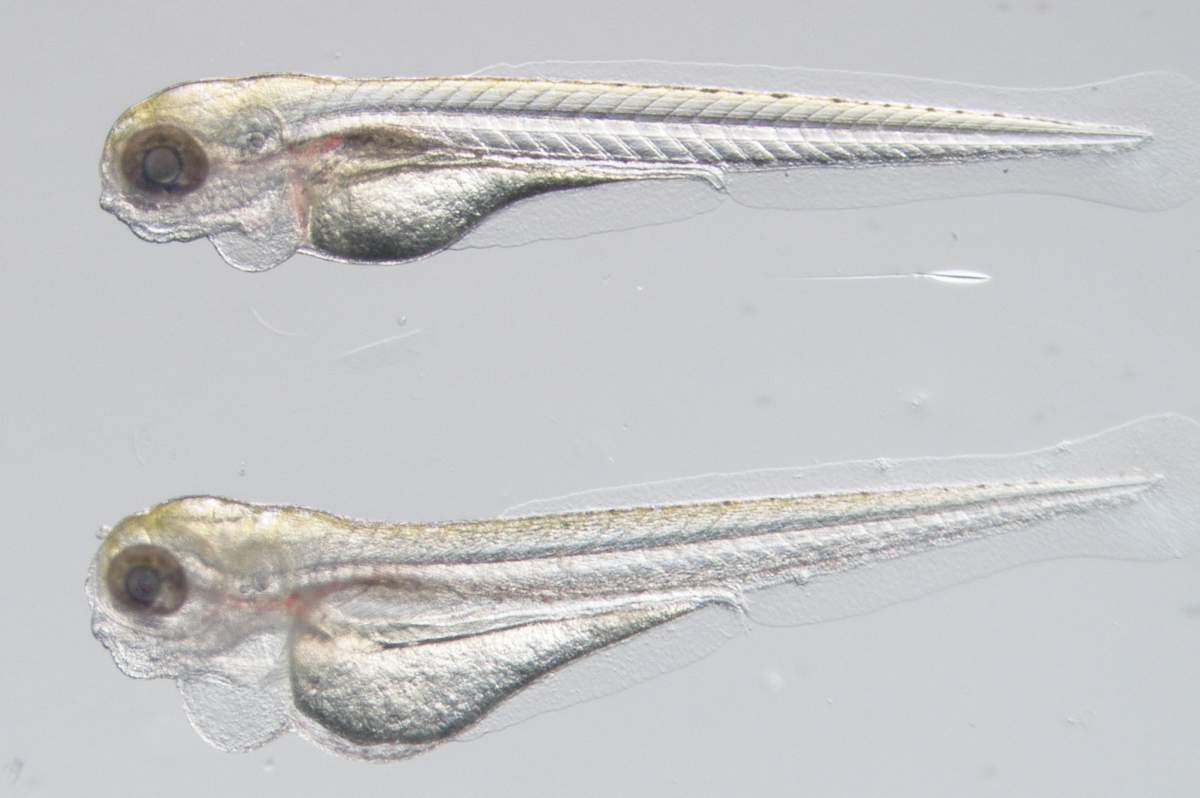
We use zebrafish, patient-derived samples, and in vitro vascular models to uncover how genetic mutations and dysregulated signaling pathways lead to abnormal vessel growth and vascular dysfunction. Our work bridges fundamental mechanisms with disease biology, including the development of the first zebrafish model of Kaposiform Lymphangiomatosis (KLA). By integrating genetic data from patients with functional testing in living models, we aim to identify therapeutic targets and develop new strategies to treat vascular and lymphatic disorders.
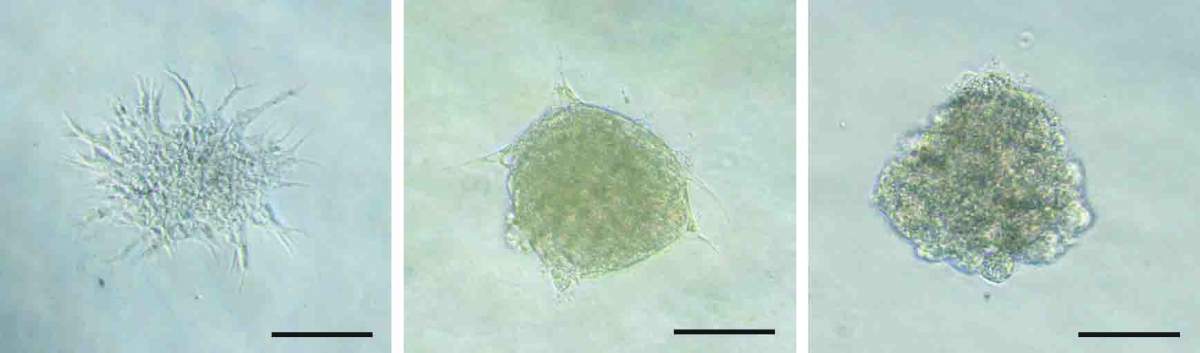
Cerebral small vessel disease- Understanding Neuro-Vascular interactions

Cerebral small vessel disease is a leading cause of stroke and cognitive decline, yet its cellular origins remain poorly understood. In our recent work, we uncovered how defects in neurovascular communication disrupt cerebral vessel growth and compromise blood-brain barrier integrity. Using zebrafish and complementary models, we showed that disrupted glial differentiation impairs brain angiogenesis and vascular stability. These findings highlight glia-vascular interactions as key regulators of brain vascular integrity and blood-brain barrier function, with important implications for cerebral small vessel disease.
The Biology Behind Venous Failure
Varicose veins are the most common form of chronic vascular disease, affecting approximately 10%-30% of the population. They are caused by venous insufficiency, which manifests in the inability of veins in the lower extremities to return blood flow to the heart against gravity. This dysfunction leads to bulging veins, which may progress to pain, edema, and even thrombosis and venous ulcers. Although the precise pathophysiology of the disease is still debatable, varicose vein development is known to involve valve incompetence, increased intravenous pressure, and inflammatory changes in the vessel wall affecting its compliance. In our project we aim to characterize the morphological, molecular, and functional changes associated with venous insufficiency and identify the genetic cues involved in varicose vein disease.
Visualizing Human Vascular Architecture in 3D

Human saphenous vein imaged using lightsheet microscopy following CD31 staining and iDISCO tissue clearing. The vessel lumen (blue) is lined by endothelial cells (light blue) forming the intima, the innermost barrier between blood flow and the vessel wall. Surrounding the vessel, the vasa vasorum (green) form a microvascular network that supplies oxygen and nutrients to the outer layers of the vessel wall. This 3D reconstruction illustrates how large vessels sustain their own microcirculatory support system.

