Publications
2025
-
(2025) Neuron. 113, 16, p. 2692-2707.e8 Abstract
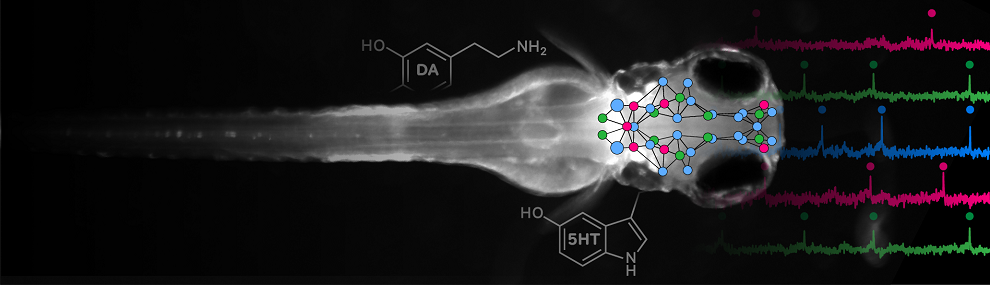
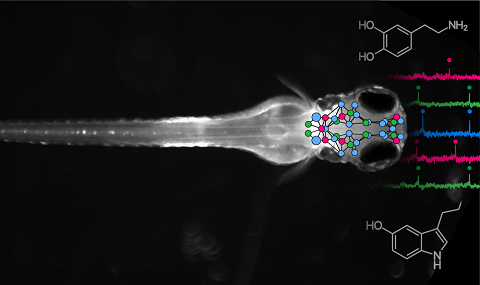
As animals adapt to new situations, neuromodulation is a potent way to alter behavior, yet mechanisms by which neuromodulatory nuclei compute during behavior are underexplored. The serotonergic raphe supports motor learning in larval zebrafish by visually detecting distance traveled during swims, encoding action effectiveness, and modulating motor vigor. We tracked the raphe's input-output computations at millisecond timescales using voltage and neurotransmitter imaging and found that swimming opens a gate for visual input to cause spiking in serotonergic neurons, enabling the encoding of action outcomes and filtering out learning-irrelevant visual signals. Specifically, swim commands initially inhibited serotonergic neurons via γ-aminobutyric acid (GABA). Immediately after, membrane voltage increased via post-inhibitory rebound, allowing swim-induced visual motion to evoke firing through glutamate, triggering serotonin release to modulate future motor vigor. Ablating local GABAergic neurons impaired raphe coding and motor learning. Thus, serotonergic neuromodulation arises from action-outcome coincidence detection within the raphe.
2024
-
(2024) BioRxiv. Abstract
The vertebrate neuromodulatory systems play a critical role in controlling adaptive behavior. Yet, it has been challenging to unravel their downstream targets due to their widespread innervation and the complex nature of receptor signaling. Here, we show that the serotonergic system controls brain-wide neural dynamics in a spatially dualistic manner, global and compartmentalized, during motor adaptation in zebrafish. Larval zebrafish adapt the vigor of tail motions depending on environmental drag force and based on sensory cues during the optomotor response in a serotonin-dependent manner. Whole-brain neural activity imaging combined with the perturbation of tph2+ raphe serotonin neurons revealed dualistic modulation of neural activity depending on behavioral encoding: global suppression of locomotor networks and compartmentalized enhancement of midbrain sensory networks, both of which synergistically enabled behavioral adaptation. Moreover, whole-brain imaging of serotonin release and systematic spatial mapping of serotonin receptors showed highly compartmentalized patterns that span multiple brain areas, which explained the compartmentalized modulation of sensory encoding. The global modulation of motor encoding occurred through a key network hub that broadcasts behavioral state signals, overriding local neuromodulation. Our results reveal how a neuromodulatory system interacts with brain-wide neural dynamics through its parallel interactions and provide a conceptual framework for understanding the neural mechanisms of neuromodulatory behavioral control.
-
(2024) Molecular Psychiatry. 29, 4, p. 1046-1062 Abstract
Serotonergic psychedelics are emerging therapeutics for psychiatric disorders, yet their underlying mechanisms of action in the brain remain largely elusive. Here, we developed a wide-field behavioral tracking system for larval zebrafish and investigated the effects of psilocybin, a psychedelic serotonin receptor agonist. Machine learning analyses of precise body kinematics identified latent behavioral states reflecting spontaneous exploration, visually-driven rapid swimming, and irregular swim patterns following stress exposure. Using this method, we found that acute psilocybin treatment has two behavioral effects: [i] facilitation of spontaneous exploration (\u201cstimulatory\u201d) and [ii] prevention of irregular swim patterns following stress exposure (\u201canxiolytic\u201d). These effects differed from the effect of acute SSRI treatment and were rather similar to the effect of ketamine treatment. Neural activity imaging in the dorsal raphe nucleus suggested that psilocybin inhibits serotonergic neurons by activating local GABAergic neurons, consistent with psychedelic-induced suppression of serotonergic neurons in mammals. These findings pave the way for using larval zebrafish to elucidate neural mechanisms underlying the behavioral effects of serotonergic psychedelics.
2023
-
(2023) Nature Computational Science. 3, 1, p. 71-85 Abstract
Calcium imaging has been widely adopted for its ability to record from large neuronal populations. To summarize the time course of neural activity, dimensionality-reduction methods, which have been applied extensively to population spiking activity, may be particularly useful. However, it is unclear whether the dimensionality-reduction methods applied to spiking activity are appropriate for calcium imaging. We thus carried out a systematic study of design choices based on standard dimensionality-reduction methods. We have also developed a method to perform deconvolution and dimensionality reduction simultaneously (calcium imaging linear dynamical system, CILDS). CILDS most accurately recovered the single-trial, low-dimensional time courses from simulated calcium imaging data. CILDS also outperformed the other methods on calcium imaging recordings from larval zebrafish and mice. More broadly, this study represents a foundation for summarizing calcium-imaging recordings of large neuronal populations using dimensionality reduction in diverse experimental settings.
2022
-
(2022) Frontiers in Cell and Developmental Biology. 10, 875044. Abstract
Understanding how neurons interact across the brain to control animal behaviors is one of the central goals in neuroscience. Recent developments in fluorescent microscopy and genetically-encoded calcium indicators led to the establishment of whole-brain imaging methods in zebrafish, which record neural activity across a brain-wide volume with single-cell resolution. Pioneering studies of whole-brain imaging used custom light-sheet microscopes, and their operation relied on commercially developed and maintained software not available globally. Hence it has been challenging to disseminate and develop the technology in the research community. Here, we present PyZebrascope, an open-source Python platform designed for neural activity imaging in zebrafish using light-sheet microscopy. PyZebrascope has intuitive user interfaces and supports essential features for whole-brain imaging, such as two orthogonal excitation beams and eye damage prevention. Its camera module can handle image data throughput of up to 800 MB/s from camera acquisition to file writing while maintaining stable CPU and memory usage. Its modular architecture allows the inclusion of advanced algorithms for microscope control and image processing. As a proof of concept, we implemented a novel automatic algorithm for maximizing the image resolution in the brain by precisely aligning the excitation beams to the image focal plane. PyZebrascope enables whole-brain neural activity imaging in fish behaving in a virtual reality environment. Thus, PyZebrascope will help disseminate and develop light-sheet microscopy techniques in the neuroscience community and advance our understanding of whole-brain neural dynamics during animal behaviors.
-
(2022) Neuron. 110, 7, p. 1211-1222 e4. Abstract
Motor systems must continuously adapt their output to maintain a desired trajectory. While the spinal circuits underlying rhythmic locomotion are well described, little is known about how the network modulates its output strength. A major challenge has been the difficulty of recording from spinal neurons during behavior. Here, we use voltage imaging to map the membrane potential of large populations of glutamatergic neurons throughout the spinal cord of the larval zebrafish during fictive swimming in a virtual environment. We characterized a previously undescribed subpopulation of tonic-spiking ventral V3 neurons whose spike rate correlated with swimming strength and bout length. Optogenetic activation of V3 neurons led to stronger swimming and longer bouts but did not affect tail beat frequency. Genetic ablation of V3 neurons led to reduced locomotor adaptation. The power of voltage imaging allowed us to identify V3 neurons as a critical driver of locomotor adaptation in zebrafish.
2019
-
(2019) Nature Methods. 16, 8, p. 763-770 Abstract
Current techniques for monitoring GABA (gamma-aminobutyric acid), the primary inhibitory neurotransmitter in vertebrates, cannot follow transients in intact neural circuits. To develop a GABA sensor, we applied the design principles used to create the fluorescent glutamate receptor iGluSnFR. We used a protein derived from a previously unsequenced Pseudomonas fluorescens strain and performed structure-guided mutagenesis and library screening to obtain intensity-based GABA sensing fluorescence reporter (iGABASnFR) variants. iGABASnFR is genetically encoded, detects GABA release evoked by electric stimulation of afferent fibers in acute brain slices and produces readily detectable fluorescence increases in vivo in mice and zebrafish. We applied iGABASnFR to track mitochondrial GABA content and its modulation by an anticonvulsant, swimming-evoked, GABA-mediated transmission in zebrafish cerebellum, GABA release events during interictal spikes and seizures in awake mice, and found that GABA-mediated tone decreases during isoflurane anesthesia.
-
(2019) Science. 365, 6454, p. 699-704 Abstract
Genetically encoded voltage indicators (GEVIs) enable monitoring of neuronal activity at high spatial and temporal resolution. However, the utility of existing GEVIs has been limited by the brightness and photostability of fluorescent proteins and rhodopsins. We engineered a GEVI, called Voltron, that uses bright and photostable synthetic dyes instead of protein-based fluorophores, thereby extending the number of neurons imaged simultaneously in vivo by a factor of 10 and enabling imaging for significantly longer durations relative to existing GEVIs. We used Voltron for in vivo voltage imaging in mice, zebrafish, and fruit flies. In the mouse cortex, Voltron allowed single-trial recording of spikes and subthreshold voltage signals from dozens of neurons simultaneously over a 15-minute period of continuous imaging. In larval zebrafish, Voltron enabled the precise correlation of spike timing with behavior.
-
(2019) eLife. 8, e44927. Abstract
Uninterrupted arousal is important for survival during threatening situations. Activation of orexin/hypocretin neurons is implicated in sustained arousal. However, orexin neurons produce and release orexin as well as several co-transmitters including dynorphin and glutamate. To disambiguate orexin-dependent and -independent physiological functions of orexin neurons, we generated a novel Orexin-flippase (Flp) knock-in mouse line. Crossing with Flp-reporter or Creexpressing mice showed gene expression exclusively in orexin neurons. Histological studies confirmed that orexin was knock-out in homozygous mice. Orexin neurons without orexin showed altered electrophysiological properties, as well as received decreased glutamatergic inputs. Selective chemogenetic activation revealed that both orexin and co-transmitters functioned to increase wakefulness, however, orexin was indispensable to promote sustained arousal. Surprisingly, such activation increased the total time spent in cataplexy. Taken together, orexin is essential to maintain basic membrane properties and input-output computation of orexin neurons, as well as to exert awake-sustaining aptitude of orexin neurons.
2018
-
(2018) Cell Reports. 25, 3, p. 640-650.e2 Abstract
Neural network remodeling underpins the ability to remember life experiences, but little is known about the long-term plasticity of neural populations. To study how the brain encodes episodic events, we used time-lapse two-photon microscopy and a fluorescent reporter of neural plasticity based on an enhanced form of the synaptic activity-responsive element (E-SARE) within the Arc promoter to track thousands of CA1 hippocampal pyramidal cells over weeks in mice that repeatedly encountered different environments. Each environment evokes characteristic patterns of ensemble neural plasticity, but with each encounter, the set of activated cells gradually evolves. After repeated exposures, the plasticity patterns evoked by an individual environment progressively stabilize. Compared with young adults, plasticity patterns in aged mice are less specific to individual environments and less stable across repeat experiences. Long-term consolidation of hippocampal plasticity patterns may support long-term memory formation, whereas weaker consolidation in aged subjects might reflect declining memory function.
-
(2018) Nature Chemical Biology. 14, 4, p. 352-360 Abstract
We developed a new way to engineer complex proteins toward multidimensional specifications using a simple, yet scalable, directed evolution strategy. By robotically picking mammalian cells that were identified, under a microscope, as expressing proteins that simultaneously exhibit several specific properties, we can screen hundreds of thousands of proteins in a library in just a few hours, evaluating each along multiple performance axes. To demonstrate the power of this approach, we created a genetically encoded fluorescent voltage indicator, simultaneously optimizing its brightness and membrane localization using our microscopy-guided cell-picking strategy. We produced the high-performance opsin-based fluorescent voltage reporter Archon1 and demonstrated its utility by imaging spiking and millivolt-scale subthreshold and synaptic activity in acute mouse brain slices and in larval zebrafish in vivo. We also measured postsynaptic responses downstream of optogenetically controlled neurons in C. elegans.
-
(2018) Neuroscience Research. 129, p. 32-39 Abstract
The serotonergic system in the vertebrate brain is implicated in various behaviors and diseases. Its involvement in motor control has been studied for over half a century, but efforts to build a unified model of its functions have been hampered due to the complexity of serotonergic neuromodulation. This review summarizes the anatomical structure of the serotonergic system, its afferent and efferent connections to other brain regions, and recent insights into the sensorimotor computations in the serotonergic system, and considers future research directions into the roles of serotonergic system in motor control.
2016
-
(2016) Cell. 167, 4, p. 933-946.e20 Abstract
To execute accurate movements, animals must continuously adapt their behavior to changes in their bodies and environments. Animals can learn changes in the relationship between their locomotor commands and the resulting distance moved, then adjust command strength to achieve a desired travel distance. It is largely unknown which circuits implement this form of motor learning, or how. Using whole-brain neuronal imaging and circuit manipulations in larval zebrafish, we discovered that the serotonergic dorsal raphe nucleus (DRN) mediates short-term locomotor learning. Serotonergic DRN neurons respond phasically to swim-induced visual motion, but little to motion that is not self-generated. During prolonged exposure to a given motosensory gain, persistent DRN activity emerges that stores the learned efficacy of motor commands and adapts future locomotor drive for tens of seconds. The DRN's ability to track the effectiveness of motor intent may constitute a computational building block for the broader functions of the serotonergic system.
2014
-
(2014) Neuron. 84, 1, p. 92-106 Abstract
CREB is a pivotal mediator of activity-regulated gene transcription that underlies memory formation and allocation. The contribution of a key CREB cofactor, CREB-regulated transcription coactivator 1 (CRTC1), has, however, remained elusive. Here we show that several constitutive kinase pathways and an activity-regulated phosphatase, calcineurin, converge to determine the nucleocytoplasmic shuttling of CRTC1. This, in turn, triggered an activity-dependent association of CRTC1 with CREB-dependent regulatory elements found on IEG promoters. Forced expression of nuclear CRTC1 in hippocampal neurons activated CREB-dependent transcription, and was sufficient to enhance contextual fear memory. Surprisingly, during contextual fear conditioning, we found evidence of nuclear recruitment of endogenous CRTC1 only in the basolateral amygdala, and not in the hippocampus. Consistently, CRTC1 knockdown in the amygdala, but not in the hippocampus, significantly attenuated fear memory. Thus, CRTC1 has a wide impact on CREB-dependent memory processes, but fine-tunes CREB output in a region-specific manner.
-
(2014) Nature Methods. 11, 9, p. 883-884 Abstract
The processing of sensory input and the generation of behavior involves large networks of neurons 1,2, which necessitates new technology 37 for recording from many neurons in behaving animals. In the larval zebrafish, light-sheet microscopy can be used to record the activity of almost all neurons in the brain simultaneously at single-cell resolution 3,4. Existing implementations, however, cannot be combined with visually driven behavior because the light sheet scans over the eye, interfering with presentation of controlled visual stimuli. Here we describe a system that overcomes the confounding eye stimulation through the use of two light sheets and combines whole-brain light-sheet imaging 3 with virtual reality for fictively behaving 1 larval zebrafish.
-
(2014) Nature Methods. 11, 9, p. 941-950 Abstract
Understanding brain function requires monitoring and interpreting the activity of large networks of neurons during behavior. Advances in recording technology are greatly increasing the size and complexity of neural data. Analyzing such data will pose a fundamental bottleneck for neuroscience. We present a library of analytical tools called Thunder built on the open-source Apache Spark platform for large-scale distributed computing. The library implements a variety of univariate and multivariate analyses with a modular, extendable structure well-suited to interactive exploration and analysis development. We demonstrate how these analyses find structure in large-scale neural data, including whole-brain light-sheet imaging data from fictively behaving larval zebrafish, and two-photon imaging data from behaving mouse. The analyses relate neuronal responses to sensory input and behavior, run in minutes or less and can be used on a private cluster or in the cloud. Our open-source framework thus holds promise for turning brain activity mapping efforts into biological insights.
-
(2014) Frontiers in Neural Circuits. 8, APR, 37. Abstract
Genetic labeling of neurons with a specific response feature is an emerging technology for precise dissection of brain circuits that are functionally heterogeneous at the single-cell level. While immediate early gene mapping has been widely used for decades to identify brain regions which are activated by external stimuli, recent characterization of the promoter and enhancer elements responsible for neuronal activity-dependent transcription have opened new avenues for live imaging of active neurons. Indeed, these advancements provided the basis for a growing repertoire of novel experiments to address the role of active neuronal networks in cognitive behaviors. In this review, we summarize the current literature on the usage and development of activity-dependent promoters and discuss the future directions of this expanding new field.
2013
-
(2013) Nature Methods. 10, 9, p. 889-895 Abstract
Identifying the neuronal ensembles that respond to specific stimuli and mapping their projection patterns in living animals are fundamental challenges in neuroscience. To this end, we engineered a synthetic promoter, the enhanced synaptic activity-responsive element (E-SARE), that drives neuronal activity-dependent gene expression more potently than other existing immediate-early gene promoters. Expression of a drug-inducible Cre recombinase downstream of E-SARE enabled imaging of neuronal populations that respond to monocular visual stimulation and tracking of their long-distance thalamocortical projections in living mice. Targeted cell-attached recordings and calcium imaging of neurons in sensory cortices revealed that E-SARE reporter expression correlates with sensory-evoked neuronal activity at the single-cell level and is highly specific to the type of stimuli presented to the animals. This activity-dependent promoter can expand the repertoire of genetic approaches for high-resolution anatomical and functional analysis of neural circuits.
2012
-
(2012) PLoS ONE. 7, 10, 46157. Abstract
We are interested in identifying and characterizing various projection neurons that constitute the neocortical circuit. For this purpose, we developed a novel lentiviral vector that carries the tetracycline transactivator (tTA) and the transgene under the TET Responsive Element promoter (TRE) on a single backbone. By pseudotyping such a vector with modified rabies G-protein, we were able to express palmitoylated-GFP (palGFP) or turboFP635 (RFP) in corticothalamic, corticocortical, and corticopontine neurons of mice. The high-level expression of the transgene achieved by the TET-Off system enabled us to observe characteristic elaboration of neuronal processes for each cell type. At higher magnification, we were able to observe fine structures such as boutons and spines as well. We also injected our retrograde TET-Off vector to the marmoset cortex and proved that it can be used to label the long-distance cortical connectivity of millimeter scale. In conclusion, our novel retrograde tracer provides an attractive option to investigate the morphologies of identified cortical projection neurons of various species.
-
Inverse Synaptic Tagging of Inactive Synapses via Dynamic Interaction of Arc/Arg3.1 with CaMKII beta(2012) Cell. 149, 4, p. 886-898 Abstract
The Arc/Arg3.1 gene product is rapidly upregulated by strong synaptic activity and critically contributes to weakening synapses by promoting AMPA-R endocytosis. However, how activity-induced Arc is redistributed and determines the synapses to be weakened remains unclear. Here, we show targeting of Arc to inactive synapses via a high-affinity interaction with CaMKII beta that is not bound to calmodulin. Synaptic Arc accumulates in inactive synapses that previously experienced strong activation and correlates with removal of surface GluA1 from individual synapses. A lack of CaMKII beta either in vitro or in vivo resulted in loss of Arc upregulation in the silenced synapses. The discovery of Arc's role in "inverse'' synaptic tagging that is specific for weaker synapses and prevents undesired enhancement of weak synapses in potentiated neurons reconciles essential roles of Arc both for the late phase of long-term plasticity and for reduction of surface AMPA-Rs in stimulated neurons.
2009
-
(2009) Proceedings of the National Academy of Sciences of the United States of America. 106, 1, p. 316-321 Abstract
The neuronal immediate early gene Arc/Arg-3.1 is widely used as one of the most reliable molecular markers for intense synaptic activity in vivo. However, the cis-acting elements responsible for such stringent activity dependence have not been firmly identified. Here we combined luciferase reporter assays in cultured cortical neurons and comparative genome mapping to identify the critical synaptic activity-responsive elements (SARE) of the Arc/Arg-3.1 gene. A major SARE was found as a unique approximate to 100-bp element located at >5 kb upstream of the Arc/Arg-3.1 transcription initiation site in the mouse genome. This single element, when positioned immediately upstream of a minimal promoter, was necessary and sufficient to replicate crucial properties of endogenous Arc/Arg-3.1's transcriptional regulation, including rapid onset of transcription triggered by synaptic activity and low basal expression during synaptic inactivity. We identified the major determinants of SARE as a unique cluster of neuronal activity-dependent cis-regulatory elements consisting of closely localized binding sites for CREB, MEF2, and SRF. Consistently, a SARE reporter could readily trace and mark an ensemble of cells that have experienced intense activity in the recent past in vivo. Taken together, our work uncovers a novel transcriptional mechanism by which a critical 100-bp element, SARE, mediates a predominant component of the synapse-to-nucleus signaling in ensembles of Arc/Arg-3.1-positive activated neurons.