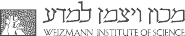Anomalous Thermal Relaxations
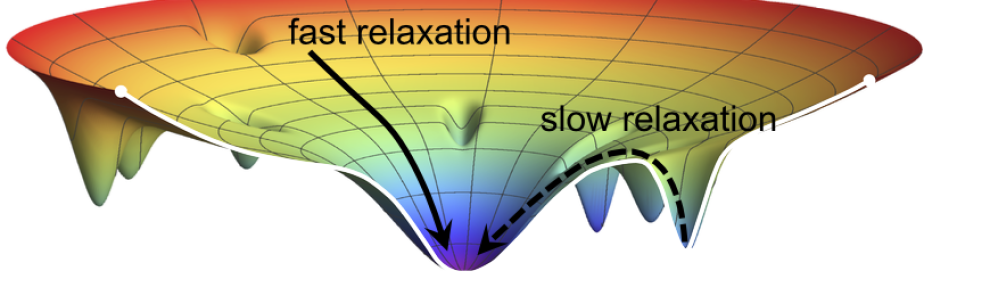
Consider two systems, identical in all macroscopic parameters except their temperatures. When both are coupled to the same environment with a different temperature (say colder or hotter than both systems), which system takes a shorter time to relax to the temperature of the environment? Surprisingly, the relaxation time is not always monotonic with the initial temperature: in some cases the initially hot system cools faster than the initially cold one, or the initially cold system heats faster than the initially hot one. These are examples of ``anomalous relaxation processes", which are not well understood yet.