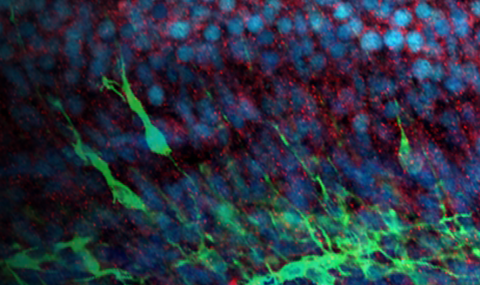
NDE1 KO organoids exhibit microlissencephaly phenotypes: Human malformations of cortical development (MCDs) are rare disorders, which have been associated with conserved brain architecture with fewer neurons (microcephaly) or associated with absent or abnormal gyri (lissencephaly). A more severe disorder is microlissencephaly, in which both the size and the folding pattern of the brain are affected, and results from a loss of function of the NDE1 gene.
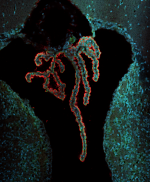
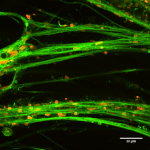
In this study, the role of NDE1 in the developing brain is investigated with different human brain organoids systems. Organoids which arise from NDE1 KO embryonic stem cells line are considerably smaller and grow slower than control organoids. In addition, in an on-chip method that allows the study of folding, the KO organoids exhibit a smoother surface than control organoids. Overall, the established NDE1 KO organoids exhibit features of microlissencephaly phenotypes.
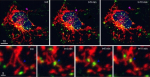
Fate change as a novel mechanism of microcephaly: To uncover the molecular variations between the NDE1 KO organoids, RNA sequencing and q-PCR experiments were performed using different organoid cultures. A shift in cell fate was observed: (1) some of the organoids expressed higher levels of genes associated with caudal brain regions such as the cerebellum and the midbrain; (2) The expression of rostral markers was reduced in most of the experiments; (3) in some organoids both the changes occurred.
The observed change in cell fate may offer insights into a novel mechanism for microcephaly. The rostral part of the neural tube, which develops into the telencephalon is widely expanded in humans, whereas the caudal regions are smaller, thus reflected in smaller organoids.