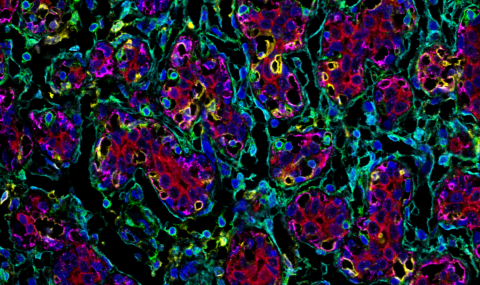
The landscape of stress responses in the tumor microenvironment
Billions of years of evolution through changing environments led organisms to develop an arsenal of cytoprotective pathways to promote their survival under stressful conditions. One such pathway is the heat shock response. We have shown that tumors can hijack the master regulator of the heat shock response, and subvert this pathway to promote their own survival, at the expense of their host. In our lab we use patient samples, mouse and tissue culture models to decipher the mechanisms by which normal cytoprotective pathways such as the heat shock response are subverted to support malignancy.