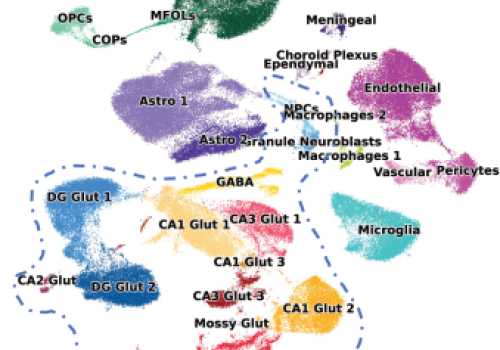
Mapping the stress response: A high-resolution single-cell atlas of the hippocampus.
Our latest publication in Molecular Psychiatry provides the most comprehensive characterization to date of the adult male hippocampus under stress, identifying cell-type specific molecular signatures.


