Warwick R. A., Riccitelli S., Heukamp A. S., Yaakov H., Swain B. P., Ankri L., Mayzel J., Gilead N., Parness-Yossifon R., Di Marco S. & Rivlin-Etzion M.
(2024)
Science advances.
10,
35,
eadk4062.
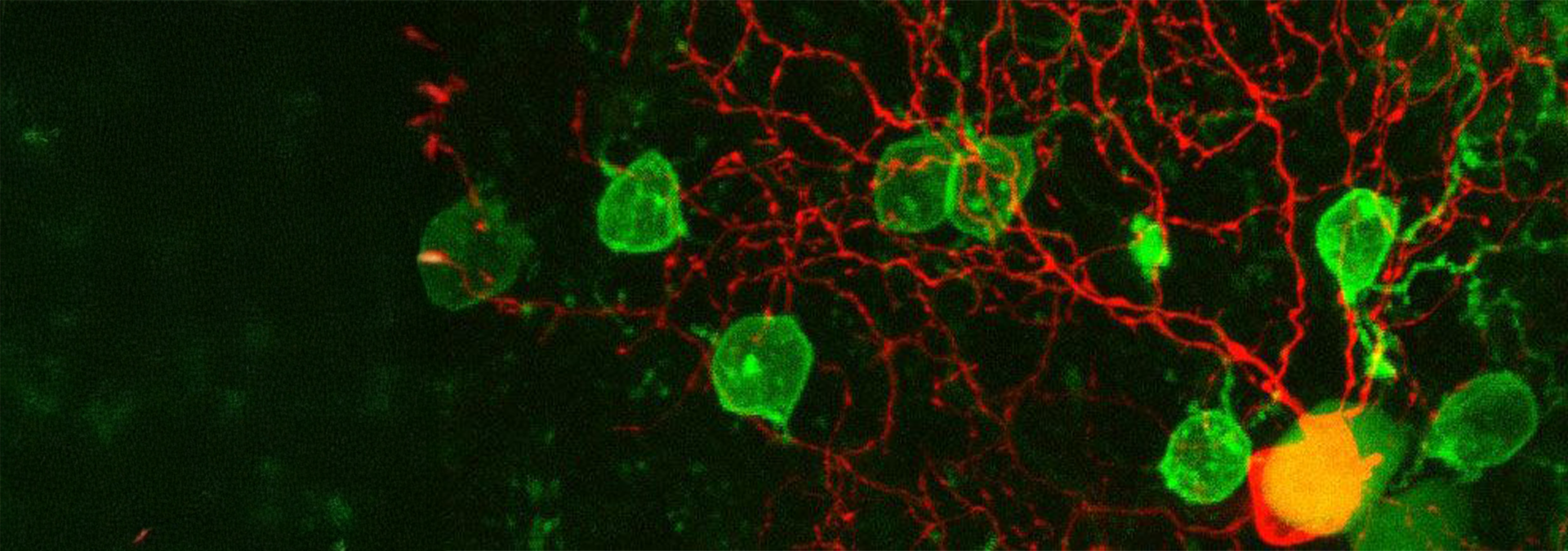
The mammalian retina is considered an autonomous circuit, yet work dating back to Ramon y Cajal indicates that it receives inputs from the brain. How such inputs affect retinal processing has remained unknown. We confirmed brain-to-retina projections of histaminergic neurons from the mouse hypothalamus. Histamine application ex vivo altered the activity of various retinal ganglion cells (RGCs), including direction-selective RGCs that gained responses to high motion velocities. These results were reproduced in vivo with optic tract recordings where histaminergic retinopetal axons were activated chemogenetically. Such changes could improve vision of fast-moving objects (e.g., while running), which fits with the known increased activity of histaminergic neurons during arousal. An antihistamine drug reduced optomotor responses to high-speed moving stimuli in freely moving mice. In humans, the same antihistamine nonuniformly modulated visual sensitivity across the visual field, indicating an evolutionary conserved function of the histaminergic system. Our findings expose a previously unappreciated role for brain-to-retina projections in modulating retinal function.