Multiplexed imaging
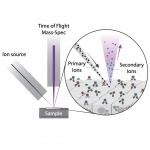
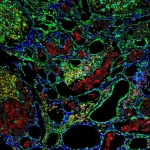
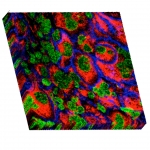
Multiplexed Ion Beam Imaging by Time of Flight (MIBI-TOF) is a new technology that enables high-dimensional single-cell imaging in situ of clinical human specimens. In MIBI-TOF, we stain human tissue sections with dozens of metal-labeled antibodies and read out their abundance and location by secondary ionization mass spectrometry. The result is a multi-dimensional image, depicting sub-cellular expression and localization for dozens of distinct proteins in situ.