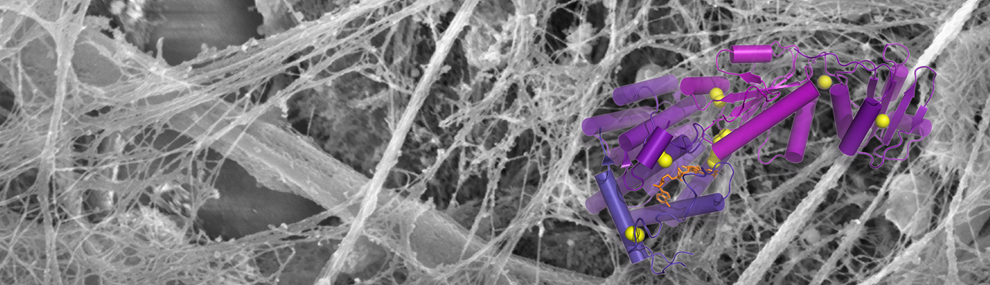
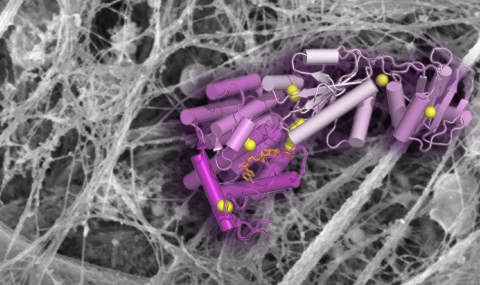
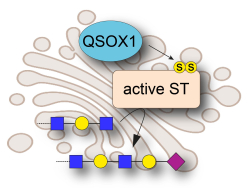
Our lab studies enzymes that catalyze the formation of disulfide bonds during the assembly of complex biological materials such as mucins and the extracellular matrix. Mucins and other disulfide-rich extracellular proteins are involved in tissue homoeostasis, protection against pathogens, and cancer. Our research combines structural and molecular biology with experiments in vivo, allowing us to determine the physiological impact of disulfide-mediated protein assembly and regulation. Controlling the activities of disulfide catalysts may have applications in medicine and tissue engineering, an avenue we are pursuing through the design of specific inhibitors of these enzymes (see movie). Our vision is that dynamic, disulfide-crosslinked, secreted network proteins are nevertheless accessible to innovative structural biological approaches and can ultimately be understood at the molecular level.