Research
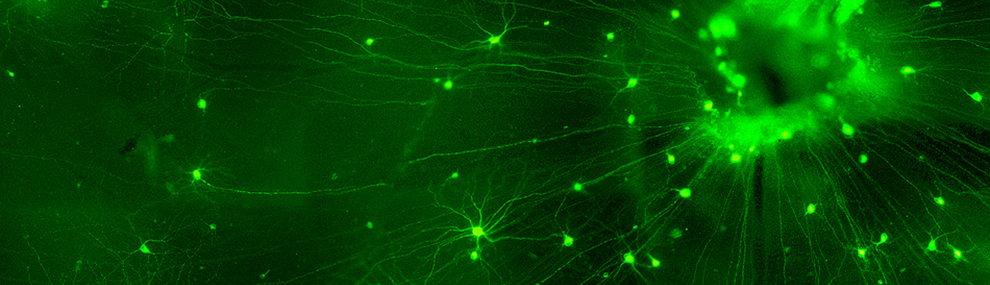
Since 1998, when we first discovered, in contrast to the prevailing dogma at the time, that innate and adaptive immune cells are needed for central nervous system protection and repair, using animal models of acute and spinal cord injuries (1, 2), the team’s focus has been on brain-immune cross talk. Subsequent to this discovery, my team demonstrated that beyond their role in repair, blood-borne immune cells support life-long brain maintenance and plasticity(3, 4), and play a key role in coping with mental stress (5, 6).
Over the years, we demonstrated that monocyte-derived macrophages promote repair by homing to the damaged CNS where they display local anti-inflammatory activity (7), and that their homing to the damaged CNS, at least in part, is via selective CNS borders, including the leptomeninges adjacent to injury site, and the remote choroid plexus epithelium within the blood cerebrospinal fluid barrier (8). (9). These studies, in addition to many others, have been instrumental in advancing our comprehension of the immune system's role in brain aging (10, 11) and neurodegenerative diseases (12). They have revealed that in the aging process (10), and in various neurodegenerative diseases involving humans and mice, there is a breakdown in the cross-talk between the brain and the immune system. This breakdown is partially attributed to the chronic expression of Type-I interferon in the brain's choroid plexus, which has a detrimental impact on brain-immune communication, impairs microglial function, and diminishes the ability to manage inflammation (10, 11, 13, 14).
Building on these findings and a plethora of other research, it has become evident that combatting chronic conditions of the brain necessitates sustained assistance from the immune system. We developed approach to enhance systemic immunity to help the brain to repair itself, by controlled blockade of the PD-1/PD-L1 inhibitory immune checkpoint (15-18). This approach is currently in clinical trial in AD patients, by ImmmunoBrain checkpoint biopharma. Our transformed understanding of brain immunity is summarized in recent articles (19-21).