Publications
2025
Temporal Genomic Analysis of Homogeneous Tumor Models Reveals Key Regulators of Immune Evasion in Melanoma
Shvefel S. C., Pai J. A., Cao Y., Pal L. R., Bartok O., Levy R., Zemanek M. J., Weller C., Herzog E., Yao W., Hiam-Galvez K. J., Cheng K., Yin Y., Du P. P., Raposo C. J., Gumpert N., Welti M., Martínez Gómez J. M., Sella F., Yakubovich E., Orr I., Ben-Dor S., Oren R., Fellus-Alyagor L., Golani O., Brenner O. J., Salame T. M., Zerbib M., Goliand I., Ranmar D., Savchenko I., Ketrarou N., Schäffer A. A., Dahan R., Levesque M. P., Ruppin E., Satpathy A. T. & Samuels Y. (2025) Cancer Discovery.
Abstract
ZEB2 is a master switch controlling the tumor-associated macrophage program
Sheban F., Phan T. S., Xie K., Ingelfinger F., Gur C., Shapir Itai Y., Blecher-Gonen R., Yu C., Avellino R., Chalan P., Freitag K., Yofe I., Yutkin V., Boyeau P., Ergen C., Hong J., Mazuz K., Liu Y., Chen K., Dahan R., Kortylewski M., Yosef N., Weiner A. & Amit I. (2025) Cancer Cell.
Abstract
CD32B1, a versatile non-signaling antibody-binding scaffold for enhanced T cell adhesion to tumor stromal cognate antigens
Feigelson S. W., Dadosh T., Levi N., Sapoznikov A., Weinstein-Marom H., Blokon-Kogan D., Avraham Y., Unger T., Gross G., Dahan R. & Alon R. (2025) Frontiers in Immunology.
Abstract
2024

Modeling T cell temporal response to cancer immunotherapy rationalizes development of combinatorial treatment protocols
Barboy O., Bercovich A., Li H., Lubling Y., Yalin A., Shapir Itai Y., Abadie K., Zada M., David E., Shlomi S., Katzenelenbogen Y., Adhemar D., Gur C., Yofe I., Feferman T., Cohen M., Dahan R., Newell E., Lifshitz A., Tanay A. & Amit I. (2024) Nature Reviews Cancer.
Abstract
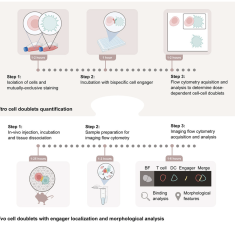
Protocol for assessing murine cell doublet engagement and subsequent effects using flow cytometry and imaging flow cytometry
Shapir Itai Y., Porat Z. & Dahan R. (2024) STAR Protocols.
Abstract
FcγRIIB is an immune checkpoint limiting the activity of Treg-targeting antibodies in the tumor microenvironment
Knorr D. A., Blanchard L., Leidner R. S., Jensen S. M., Meng R., Jones A., Ballesteros-Merino C., Bell R. B., Baez M., Marino A., Sprott D., Bifulco C. B., Piening B., Dahan R., Osorio J. C., Fox B. A. & Ravetch J. V. (2024) Cancer Immunology Research.
Abstract
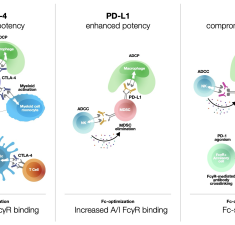
Fc-optimized checkpoint antibodies for cancer immunotherapy
Dahan R. & Korman A. J. (2024) Trends in Molecular Medicine.
Abstract
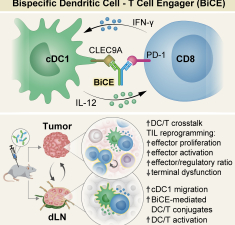
Bispecific dendritic-T cell engager potentiates anti-tumor immunity
Shapir Itai Y., Barboy O., Salomon R., Bercovich A., Xie K., Winter E., Shami T., Porat Z., Erez N., Tanay A., Amit I. & Dahan R. (2024) Cell.
Abstract
2023

Fc glycoengineering of a PD-L1 antibody harnesses Fcγ receptors for increased antitumor efficacy
Saban N. C., Yalin A., Landsberger T., Salomon R., Alva A., Feferman T., Amit I. & Dahan R. (2023) Science immunology.
Abstract
2022

Therapeutic antibody activation of the glucocorticoid-induced TNF receptor by a clustering mechanism
He C., Maniyar R. R., Avraham Y., Zappasodi R., Rusinova R., Newman W., Heath H., Wolchok J. D., Dahan R., Merghoub T. & Meyerson J. (2022) Science advances.
Abstract
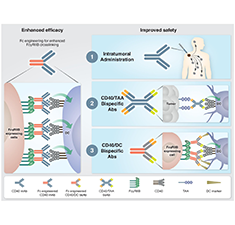
Next Generation CD40 Agonistic Antibodies for Cancer Immunotherapy
Salomon R. & Dahan R. (2022) Frontiers in Immunology.
Abstract
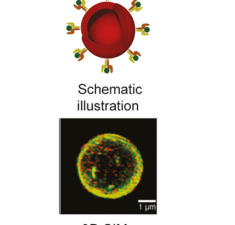
Artificial Antigen Presenting Cells for Detection and Desensitization of Autoreactive T cells Associated with Type 1 Diabetes
Artzy-Schnirman A., Abu-Shah E., Chandrawati R., Altman E., Yusuf N., Wang S. T., Ramos J., Hansel C. S., Haus-Cohen M., Dahan R., Arif S., Dustin M. L., Peakman M., Reiter Y. & Stevens M. M. (2022) Nano Letters.
Abstract

Bispecific antibodies increase the therapeutic window of CD40 agonists through selective dendritic cell targeting
Salomon R., Rotem H., Katzenelenbogen Y., Weiner A., Cohen Saban N., Feferman T., Amit I. & Dahan R. (2022) Nature Reviews Cancer.
Abstract
2021
Human antibodies targeting a Mycobacterium transporter protein mediate protection against tuberculosis
Watson A., Li H., Ma B., Weiss R., Bendayan D., Abramovitz L., Ben-Shalom N., Mor M., Pinko E., Bar Oz M., Wang Z., Du F., Lu Y., Rybniker J., Dahan R., Huang H., Barkan D., Xiang Y., Javid B. & Freund N. T. (2021) Nature Communications.
Abstract
Unified platform for genetic and serological detection of COVID-19 with single-molecule technology
Furth N., Shilo S., Cohen N., Erez N., Fedyuk V., Schrager A. M., Weinberger A., Dror A. A., Zigron A., Shehadeh M., Sela E., Srouji S., Amit S., Levy I., Segal E., Dahan R., Jones D., Douek D. C. & Shema E. (2021) PLoS ONE.
Abstract

Anti-SARS-CoV-2 antibodies elicited by COVID-19 mRNA vaccine exhibit a unique glycosylation pattern
Farkash I., Feferman T., Cohen-Saban N., Avraham Y., Morgenstern D., Mayuni G., Barth N., Lustig Y., Miller L., Shouval D. S., Biber A., Kirgner I., Levin Y. & Dahan R. (2021) Cell Reports.
Abstract
2020

Single-cell genomic approaches for developing the next generation of immunotherapies
Yofe I., Dahan R. & Amit I. (2020) Nature Medicine.
Abstract
2019
BCR affinity differentially regulates colonization of the subepithelial dome and infiltration into germinal centers within Peyer's patches
Biram A., Stromberg A., Winter E., Stoler-Barak L., Salomon R., Addadi Y., Dahan R., Yaari G., Bemark M. & Shulman Z. (2019) Nature Immunology.
Abstract
2018
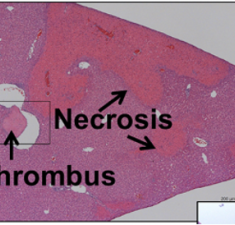
Toxicity of an Fc-engineered anti-CD40 antibody is abrogated by intratumoral injection and results in durable antitumor immunity
Knorr D. A., Dahan R. & Ravetch J. (2018) Proceedings Of The National Academy Of Sciences Of The United States Of America-Physical Sciences.
Abstract
2017
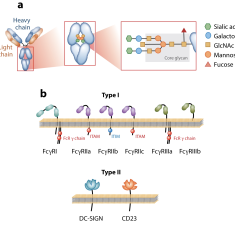
Signaling by antibodies: Recent progress
Bournazos S., Wang T. T., Dahan R., Maamary J. & Ravetch J. V. (2017) ANNUAL REVIEW OF IMMUNOLOGY.
Abstract
Fc-Optimized Anti-CD25 Depletes Tumor-Infiltrating Regulatory T Cells and Synergizes with PD-1 Blockade to Eradicate Established Tumors
Arce Vargas F., Furness A. J., Solomon I., Joshi K., Mekkaoui L., Lesko M. H., Miranda Rota E., Dahan R., Georgiou A., Sledzinska A., Ben Aissa A., Franz D., Werner Sunderland M., Wong Y. N. S., Henry J. Y., O'Brien T., Nicol D., Challacombe B., Beers S. A. & Spain L. (2017) Immunity.
Abstract
2016
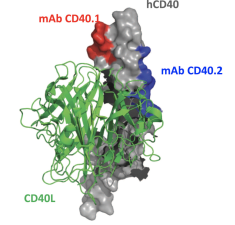
Therapeutic Activity of Agonistic, Human Anti-CD40 Monoclonal Antibodies Requires Selective FcγR Engagement
Dahan R., Barnhart B. C., Li F., Yamniuk A. P., Korman A. J. & Ravetch J. V. (2016) Cancer Cell.
Abstract
Reprogramming Tumor-Associated Macrophages by Antibody Targeting Inhibits Cancer Progression and Metastasis
Georgoudaki A. M., Prokopec K. E., Boura V. F., Hellqvist E., Sohn S., Östling J., Dahan R., Harris R. A., Rantalainen M., Klevebring D., Sund M., Brage S. E., Fuxe J., Rolny C., Li F., Ravetch J. V. & Karlsson M. C. (2016) Cell Reports.
Abstract
Co-targeting of Adenosine Signaling Pathways for Immunotherapy: Potentiation by Fc Receptor Engagement
Dahan R. & Ravetch J. V. (2016) Cancer Cell.
Abstract
2015
Erratum to FcγRs Modulate the Anti-tumor Activity of Antibodies Targeting the PD-1/PD-L1 Axis [Cancer Cell 28, 285-295; September 14, 2015]
Dahan R., Sega E., Engelhardt J., Selby M., Korman A. J. & Ravetch J. V. (2015) Cancer Cell.
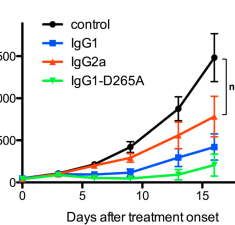
FcγRs Modulate the Anti-tumor Activity of Antibodies Targeting the PD-1/PD-L1 Axis
Dahan R., Sega E., Engelhardt J., Selby M., Korman A. J. & Ravetch J. V. (2015) Cancer Cell.
Abstract
2014
HLA-DRα1 constructs block CD74 expression and MIF effects in experimental autoimmune encephalomyelitis
Meza-Romero R., Benedek G., Yu X., Mooney J. L., Dahan R., Duvshani N., Bucala R., Offner H., Reiter Y., Burrows G. G. & Vandenbark A. A. (2014) Journal of Immunology.
Abstract
Type i and type II Fc receptors regulate innate and adaptive immunity
Pincetic A., Bournazos S., Dilillo D. J., Maamary J., Wang T. T., Dahan R., Fiebiger B. M. & Ravetch J. V. (2014) Nature Immunology.
Abstract
2013
A novel regulatory pathway for autoimmune disease: Binding of partial MHC class II constructs to monocytes reduces CD74 expression and induces both specific and bystander T-cell tolerance
Vandenbark A. A., Meza-Romero R., Benedek G., Andrew S., Huan J., Chou Y. K., Buenafe A. C., Dahan R., Reiter Y., Mooney J. L., Offner H. & Burrows G. G. (2013) Journal of Autoimmunity.
Abstract
Antigen-specific immunomodulation for type 1 diabetes by novel recombinant antibodies directed against diabetes-associates auto-reactive T cell epitope
Dahan R., Gebe J. A., Preisinger A., James E. A., Tendler M., Nepom G. T. & Reiter Y. (2013) Journal of Autoimmunity.
Abstract
2012
T-cell-receptor-like antibodies - Generation, function and applications
Dahan R. & Reiter Y. (2012) Expert Reviews in Molecular Medicine.
Abstract
2011
TCR-like antibodies distinguish conformational and functional differences in two- versus four-domain auto reactive MHC class II-peptide complexes
Dahan R., Tabul M., Chou Y. K., Meza-Romero R., Andrew S., Ferro A. J., Burrows G. G., Offner H., Vandenbark A. A. & Reiter Y. (2011) European Journal of Immunology.
Abstract
2008
Analysis of Strand Transfer and Template Switching Mechanisms of DNA Gap Repair by Homologous Recombination in Escherichia coli: Predominance of Strand Transfer
Izhar L., Goldsmith M., Dahan R., Geacintov N., Lloyd R. G. & Livneh Z. (2008) Journal of Molecular Biology.
Abstract
2007
A promising therapeutic approach for multiple sclerosis: Recombinant T-cell receptor ligands modulate experimental autoimmune encephalomyelitis by reducing interleukin-17 production and inhibiting migration of encephalitogenic cells into the CNS
Sinha S., Subramanian S., Proctor T. M., Kaler L. J., Grafe M., Dahan R., Huan J., Vandenbark A. A., Burrows G. G. & Offner H. (2007) Journal of Neuroscience.
Abstract
2004
HLA-DRB1*1501 risk association in multiple sclerosis may not be related to presentation of myelin epitopes
Finn T., Jones R., Rich C., Dahan R., Link J., David C., Chou Y., Offner H. & Vandenbark A. (2004) Journal of Neuroscience Research.
Abstract



