Faigenbaum-Romm R., Yedidi N., Gefen O., Katsowich-Nagar N., Aroeti L., Ronin I., Bar-Meir M., Rosenshine I. & Balaban N. Q.
(2025)
Cell.
188,
19,
p. 5313-5331.e18
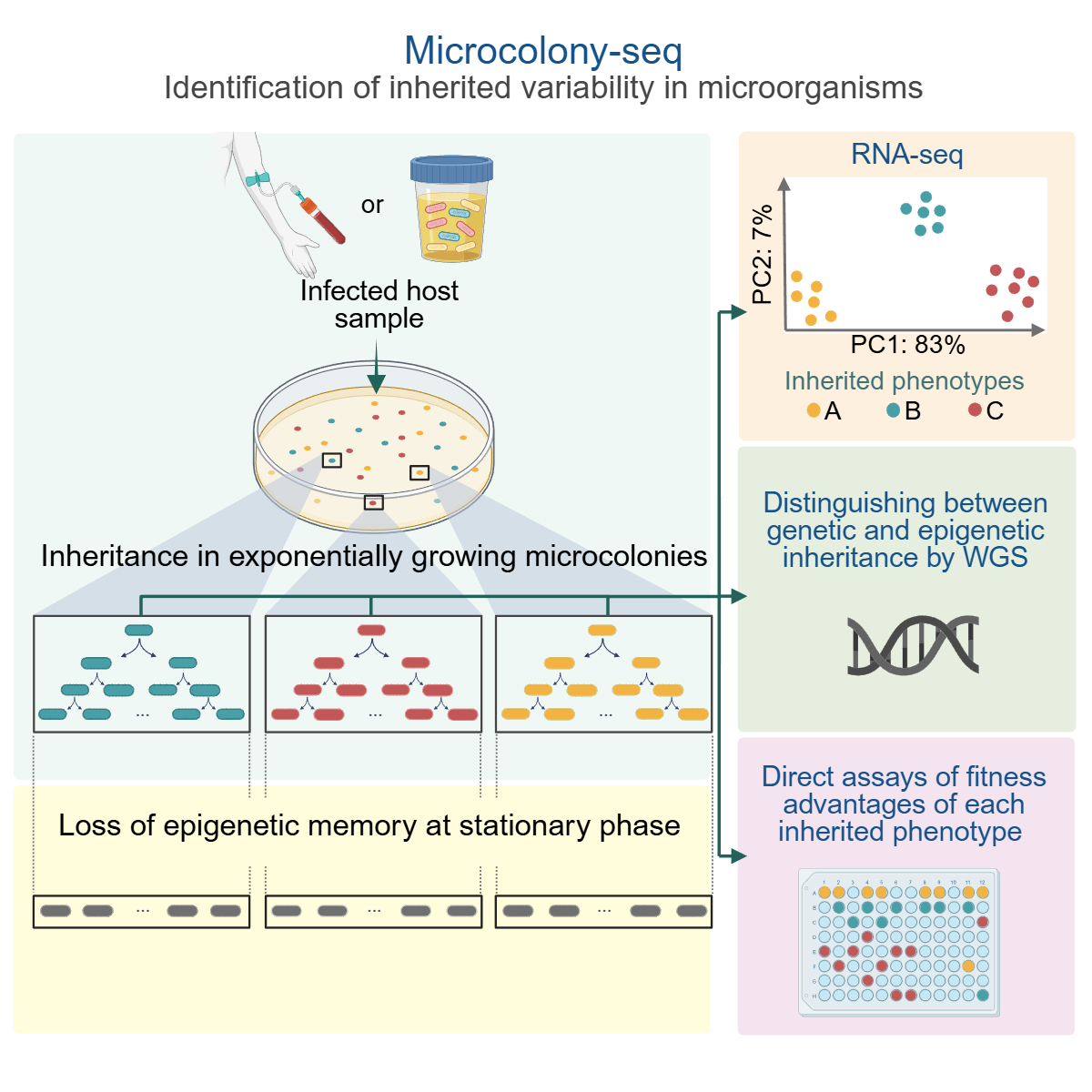
Uncovering phenotypic heterogeneity is fundamental to understanding processes such as development and stress responses. Due to the low mRNA abundance in single bacteria, determining biologically relevant heterogeneity remains a challenge. Using Microcolony-seq, a methodology that captures inherited heterogeneity by analyzing microcolonies originating from single bacterial cells, we uncover the ubiquitous ability of bacteria to maintain long-term inheritance of the host environment. Notably, we observe that growth to stationary phase erases the epigenetic inheritance. By leveraging this memory within each microcolony, Microcolony-seq combines bulk RNA sequencing (RNA-seq) with whole-genome sequencing and phenotypic assays to detect the distinct subpopulations and their fitness advantages. Applying this directly to infected human samples enables us to uncover a wealth of diverse inherited phenotypes. Our observations suggest that bacterial memory may be a widespread phenomenon in both Gram-negative and Gram-positive bacteria. Microcolony-seq provides potential targets for the rational design of therapies with the power to simultaneously target the coexisting subpopulations.