Matrix-assisted laser desorption/ionization (MALDI) imaging
Introduction
Matrix-assisted laser desorption/ionization (MALDI) employs a laser to desorb and ionize analytes. A typical MALDI imaging experiment is conducted by coating a thin tissue section mounted onto a target with an appropriate matrix solution. This solution serves to extract analytes of interest from the underlying tissue and upon solvent evaporation the extracted molecules are co-crystallized with the matrix. The role of the matrix is to absorb the laser energy and facilitate desorption/ionization of the analyte molecules. MALDI imaging is performed across the tissue at defined geometrical coordinates. Analytes present at each measurement coordinate are desorbed and ionized, and a mass spectrum is thus recorded along with its current location. Several thousand mass spectra are typically acquired in this manner. After measurement, custom software is then used to compile the mass spectra into a format where each spectrum represents a discrete pixel and the distribution and intensity of any of the detected species can be viewed across the tissue as an ion density map or image. The spatial resolution of MALDI imaging typically ranges from 5 to 100 µm.
In a single MALDI imaging experiment it is possible to detect hundreds discrete signals across a tissue from a diverse set of analytes. These analytes can range from endogenous biomolecules (e.g., sugars, organic acid, and lipids) to exogenous molecules (e.g., pharmaceutical compounds). Thus, one of the major advantages of using MALDI imaging is that endogenous biomolecules are also automatically detected, providing opportunities to evaluate the underlying physiological state of a tissue.
- MALDI profiling: acquisition of mass spectra and mass fragmentation spectra from a spotted and dried chemical standard solution or biological sample (e.g. tissue section).
- MALDI imaging: 2D imaging of biological tissues or tissue sections with a resolution down to 5µm pixel size.
- MALDI imaging with ion mobility: 2D imaging or MS profiling with separation of ions in the ion mobility cell (for ions that cannot be resolved in the MS).
- MALDI-2 post ionization: post-ionization of neutral particles desorbed by laser 1, can be used in combination with all three analysis options above.
Secondary ion mass spectrometry (SIMS) imaging
Introduction
Our state of the art instrument enables the analysis of biological samples with the lateral resolution previously only archived with TOF-SIMS technology with ultra-high mass resolution using an OrbitrapTM mass analyzer in combination with an Argon gas cluster source for the generation of secondary ions.
The following table compares MALDI imaging to traditional TOF-SIMS and Orbi-SIMS technology. Comparing TOF-SIMS and MALDI-imaging, the differences between the different kinds of analysis become obvious.
Table 1: Comparison of MALDI-MS, Orbi-SIMS and TOF-SIMS metabolite imaging techniques
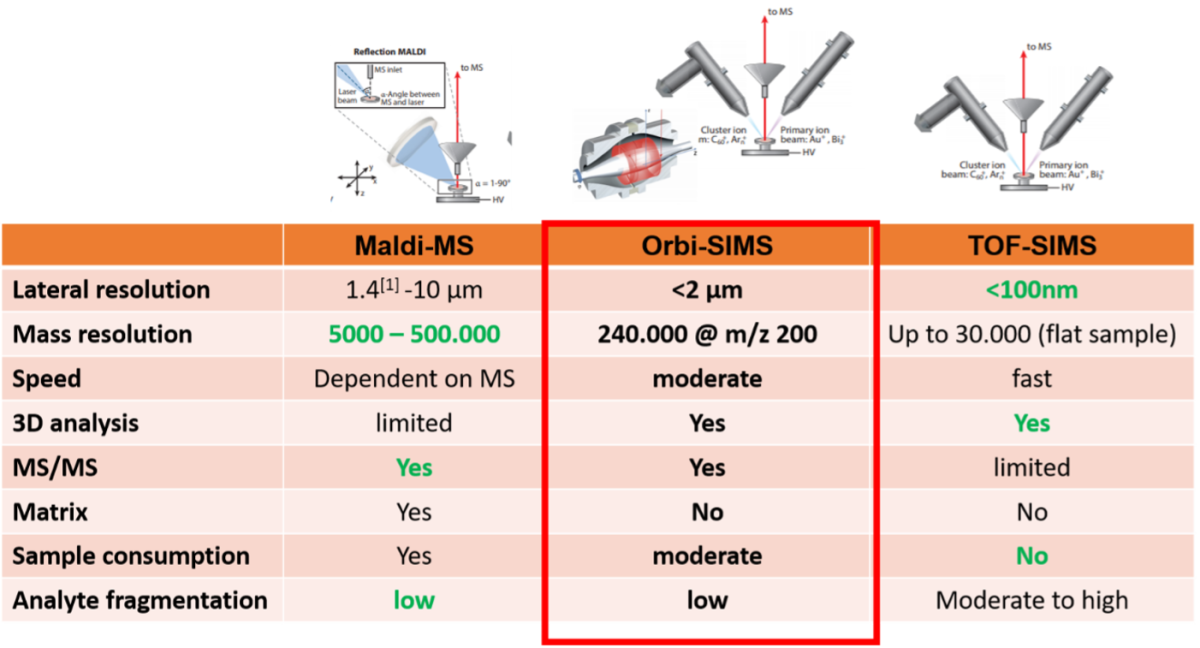
While the lateral resolution of MALDI-imaging (MSI) is limited by the laser diameter coupling to high resolution mass spectrometers, in example FT-ICR MS enables high mass resolution and molecules are not fragmented in this ionization technique. However, due to the necessity of Matrix application, MSI has limited capability for 3-dimensional metabolite detection and visualization. In contraire TOF-SIMS is extremely fast and a very high lateral resolution of <100nm can be achieved. Since it is a matrix-free technology and typically, a TOF-SIMS set-up consists of two ion-guns, one for analysis and one for sputtering, 3D acquisition is possible. The clear disadvantage of TOF-SIMS is the relatively low mass resolution and the high energy that is applied to the sample for the generation of secondary ions, what leads to high fragmentation of analyte molecules. The Orbi-SIMS technology, and especially combination of Orbi-SIMS and TOF-SIMS, like in the M6-hybrid SIMS overcomes these limitations by using the Ar-sputter beam as a ionization source with lower energy, leading to secondary ions with higher masses and less fragmentation of the compounds. The OrbitrapTM analyzer allows mass detection at a resolution of 240.000 @ m/z 200. Although due to the lower speed of the OrbitrapTM analyzer, lateral resolution is lower, combination of TOF and OrbitrapTM analyzers allow high-mass-resolution measurements with nm lateral resolution.
Scan modes of M6-hybrid SIMS
The configuration of the Weizmann-M6-hybrid SIMS instrument consists of the M6-TOF-SIMS, the nanoprobe50-liquid metal ion gun, the Argon-gas-cluster source, the QExactiveTM HF high resolution OrbitrapTM analyzer, the H/C (heating/cooling) loadlock and a dock for the Leica VCT500 cryo-shuttle. The H/C loadlock enables measurement of samples from ambient temperature to cryo-conditions (liquid nitrogen). Samples can be prepared using high-pressure freezing devices and transferred with the shuttle to the instrument.
This configuration allows measurement in all scan-modes described for the Orbi-SIMS prototype (Passarelli et. al. Nature Methods, 2017), which are illustrated in figure 1.

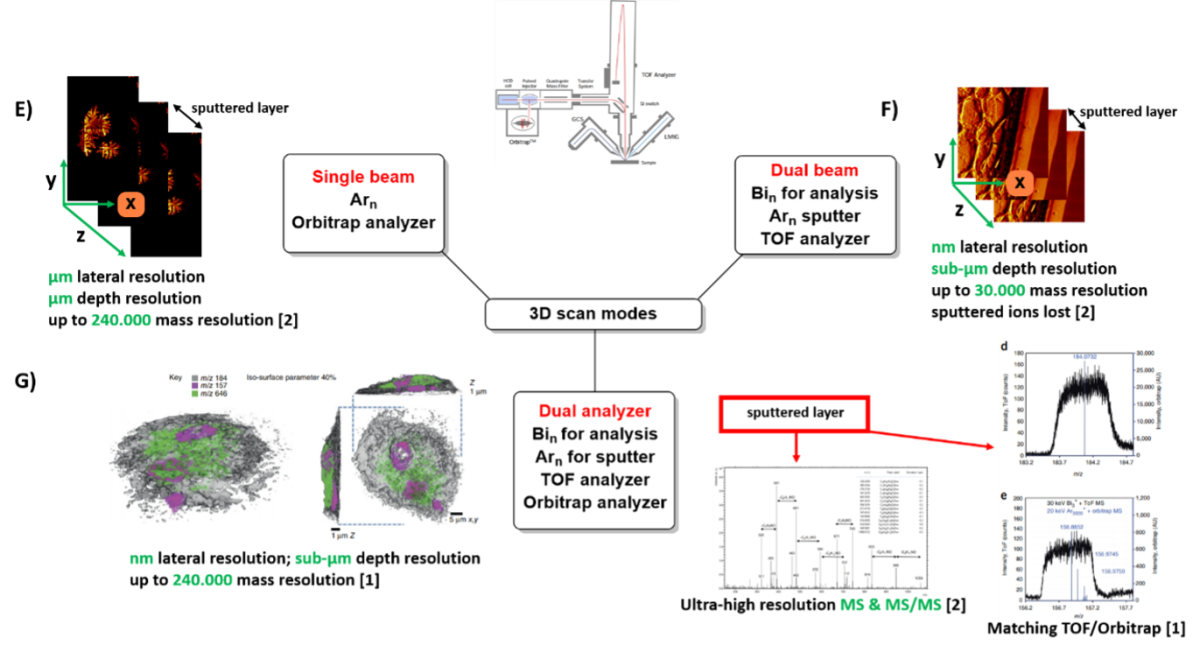
Figure 1: Scan modes of M6-hybrid-SIMS. A)-D) 1D & 2D-measurement options, E)-G) 3D measurement options. A) Acquisition of surface MS and MS/MS spectra, B) depth profiling, C) 2D-imaging with TOF analyzer and Bi-ion gun, D) 2D-imaging with OrbitrapTM analyzer and Ar-gas-cluster-source, E) single beam 3D-imaging, Ar-clusters are used for secondary ion generation and sputtering, detection with OrbitrapTM analyzer, F) dual beam 3D-imaging with Bi-clusters for secondary ion generation for analysis and Ar-clusters for sputtering, G) dual analyzer mode: Bi-cluster for secondary ion generation for analysis, Ar-clusters for sputtering, TOF analyzer for analysis of surface-sections, sputtered ions are analyzed in OrbitrapTM analyzer to obtain ultra-high mass resolution or MS/MS information. [1] Passarelli et al. Nature Methods, 2017. [2] Spectra and pictures from demo measurements at IonTOF GmbH, Münster, Germany. [3] University of Nottingham, with permission of IonTOF GmbH, Münster, Germany.
Time of flight SIMS analysis:
- 2D imaging of maximum 500x500 µm field of measurement with maximum resolution of 2048x2048 pixels (smaller areas possible, resulting in higher resolution). Minimum spot size 50nm.
- 2D large area scans: imaging of larger area (dependent on sample size) by acquisition with 5 µm resolution.
- Dual beam depth profiling using the secondary ion beam to sputter a sample layer by layer and measure the z-distribution of molecules (only organic materials).
- Dual beam 3D imaging using the secondary ion beam to sputter a sample layer by layer and acquire images in each z, resulting in a 3D image (only organic materials).
Orbitrap SIMS analysis:
- 2D imaging of maximum 500x500 µm with minimum spot size of 2 µm and mass resolution of 240.000 @ m/z 200.
- 2D large area scans: imaging of larger area (dependent on sample size) by acquisition with minimum spot size of 20 µm resolution and mass resolution of 240.000 @ m/z 200.
- Single beam depth profiling using the Ar gas cluster ion beam to simultaneously sputter a sample layer by layer and acquire spectra with the Orbitrap analyzer, resulting in depth distribution of molecules.
